ORIGINAL ARTICLE
ALENCAR FILHO, João Reginaldo de [1]
ALENCAR FILHO, João Reginaldo de. Reverse Logistics for Medicines, Supplies and Related Products, Application, and Impacts on the Public Sector. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year 05, Ed. 11, Vol. 15, pp. 33-43. November 2020. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/health/supplies-and-related-products
SUMMARY
This study aims to discuss the results of logistics management of supplements and finished products, focused on the area of medicines and correrelated in transport, storage sites, and distribution in the public sector and its subsequent impact on public health. For the accomplishment of the present study, information produced in reports in the period from 2010 to 2017 was used, in addition to consultations with data published in journals, books, articles, and electronic journals. This article also intends to present the difficulties in view of the apparent lack of better public administration and management of the drug logistics production chain, describing the activities and their challenges through the results. It is notorious that there is a large deficit in pharmaceutical logistics and service management undertaken by the public sector, in order to make the elaboration of a project that will meet the needs of the sector, with regard to the improvement of services, through the action of qualified employees, who can act in administrative processes, such as management and procurement requests in order to provide transparency and efficiency. This can be satisfied with the implementation of a management and logistics management more active and responsible in the implementation of operational processes until the delivery of products to the final consumer.
Keywords: Drug logistics, reverse logistics, Public sector, Public health, Environment.
INTRODUCTION
This research aims to expose, through data and documents, that demonstrate how public resources are used and the importance of the administrative and operational process of reverse logistics of the pharmaceutical sector in the service to the public consumer sector, aiming to ensure public health, avoid damage to health to the environment.
The use of reverse logistics of medicines and correlates aims to receive the discarded products, either by the final consumer, or by public or private entities, in order to ensure the preservation of the environment and public health. Thus, it controls the disposal and the correct destination of the garbage, which would be the responsibility of the laboratories that produce them.
The quality in the operations of receiving, storing, distributing, moving and acquiring these specific products generate the movement of billions of reais and has its commercial operations of products, such as medicines, insumos and correcorrereports of the health segment, which tries to keep up with the growth of the population and that of technology. Today consumption is increasingly aggressive, due to the accessibility of both information and access to various advertising campaigns to generate sales. All this flow ends up generating a large disposal of waste and garbage from this consumption (ANVISA, 2010).
The risk to the environment, due to the great consumption of the population, either due to lack of education and/or ignorance of how to dispose of waste, ends up polluting streets and nature itself, which ends up causing several diseases. This ends up generating great expenses for the public administration, which as a consequence will need to treat the population with public health services, in addition to drug costs.
At the end of this process, the lack of professionals specialized in administration and logistics to control the purchase, distribution and conservation of these drugs also generate major problems, usually management problems, such as the lack and control of the expiration of medicines and medicines (AGÊNCIA BRASIL, 2011).
DEVELOPMENT
According to the Ministry of the Environment, reverse logistics “is one of the tools for applying shared responsibility for the life cycle of products” (MMA, 2020.). To this end, in 2010, Law No. 12,305 of August 2, regulated by Decree No. 7,404 of 12.23.2010, was issued, complementing the arts. 16 and 17, by Decree No. 9,177, of 23 of 10, 2017, whose actions were issued by the National Solid Waste Policy (PNRS, 2010) in the country.
Also, according to the Ministry of the Environment, the PNRS defined reverse logistics as a
an instrument of economic and social development characterized by a set of actions, procedures and means aimed at enabling the collection and restitution of solid waste to the business sector, for reuse, in its cycle or in other production cycles, or other environmentally appropriate final destination. (MMA, 2020).
In simple language, it is equivalent to say that reverse logistics becomes an instrument for the control of garbage generated by various public or private institutions when the production and marketing of products end up becoming garbage, after use by potential consumers.
In the segment of medicines as many others, a set of complex operations relevant to the organization of cargo and unloading transport, such as storage and distributions to the bases of health support and maintenance of numerous deposits for the allocation of products and materials, must follow a strict quality system in operations, in addition to the importance of all managers , and who is involved in the operational process, have training and qualification, in order to ensure the satisfaction of the requirements within the Brazilian public health network.
In administrative and operational logistics in public health services, for the acquisition activity, the sector needs to know the exact demand to meet the needs of the locality, thus ensuring that its stock is always up to date and with a positive shelf life. This is essential for the manager to work, not only with a margin of security, but will also provide a balanced interaction between sectors, in relation to exchanges and transfers.
The movement, inside and outside the warehouse or factory, must ensure the safety and quality of the products and services provided, to be performed with equipment and EPIS.
The storage of products and insums shall ensure that products and logistics facilities are in accordance with the warranty and conservation of the products.
In distribution; every shipping process must meet the rules of transport, with trucks that meet the requirements to ensure the quality of the product. The entire distribution stage must be provided by qualified and qualified personnel, in order to ensure a satisfactory movement in the operationalization of the service.
National logistics in public health services faces a great lack of qualified and trained professionals to meet the needs of the various locations in Brazil, whether at the state, municipal or district level, who require the service of drug logistics and reverse logistics of medicines.
The Unified Health System (SUS, 1988) was regulated with the implementation of the Federal Constitution on October 5, 1988, to ensure that health is the right of all and the duty of the State, whose principles are based on government actions submitted to social control and whose cost is due to social security resources (33%), in the three spheres of government , with the participation of the private sector on a complementary and public rules. (CEBES, 2010).
Added another decade later, it is concluded that the public health sector still lives in endless crises, which undermine public resources, public health and the environment.
One of the factors that contributes to this reality and that is intended to be highlighted here about the professionals of the sector, who have little or no qualification to act in the operational responsibilities that demand the health area related to medicines and medical products. This unpreparedness in skilled labor can end up culminating in errors and risks to the health of people who need to use them and who are not aware of how to dispose of garbage from these products, thus negatively affecting the environment.
A qualified professional in the process of purchasing products and insums, besides being qualified, would need to have some experience, to be able to evaluate the state of demand to make a conscious purchase, and operationalize the services related to the space for storage and procedure of movement of products and insums.
The planning that managers of distribution centers and hospitals with large circulation of people and products, are not always based on modern processes and systems that would allow agility in care and quality control. This makes it difficult to process and control the products, due to lack of planning. The current Logistics Evolutionary Cycle begins in planning, from studies on demand, to the need and quantity of certain products or insums.
Figure 1. Logistics Evolutionary Cycle
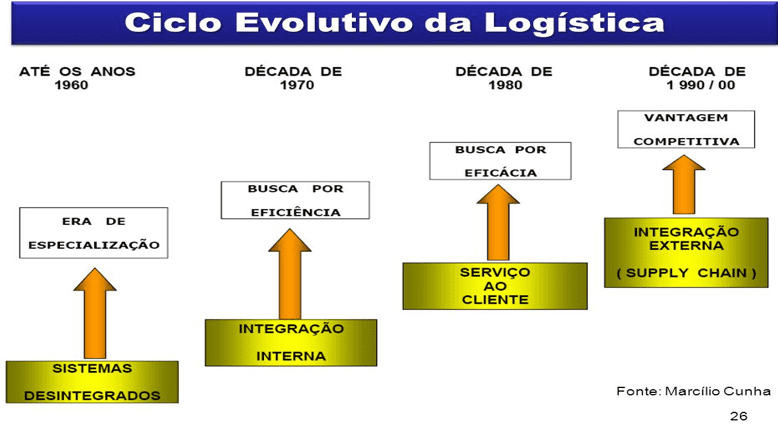
Source: Marcilo Cunha, 26
In health companies or institutions, it is of the utmost importance that people involved in the activities of supplying medical products and supplies have the ability to perform the service so as not to cause damage from the receipt, management and management of these materials.
In the case of hospital institutions, logistics studies the seasonality of products, to strategically seek the best suppliers and best prices, in order to prevent waste. With this it can ensure efficiency in the operational process and quality in this activity.
In order to avoid errors and damage to people’s health and the environment, reverse logistics is supported by Law 12,305, of 8/2/2010, regulated by Decrees 7,404 of 12/23/2010 and 9,177 of 10/23 / 2017, which provides, among other things, the implementation of the reverse logistics system. (MMA, 2020).
In addition to regulation, the law stipulates two other instruments to enable reverse logistics: the term of commitment and the sectoral agreement.
Figure 2. Decree establishes the reverse logistics system of medicines
According to the Federal Constitution of 1988, the obligation to bid seeks the demonstration of efficiency, transparency and morality in administrative business, preventing members of the administration, in its circle of personal or family friendships, from benefiting in some way, even if it is in favor of the Public Power. (According to Article 3 of Law 8666/93).
Figure 3. Reverse Logistics
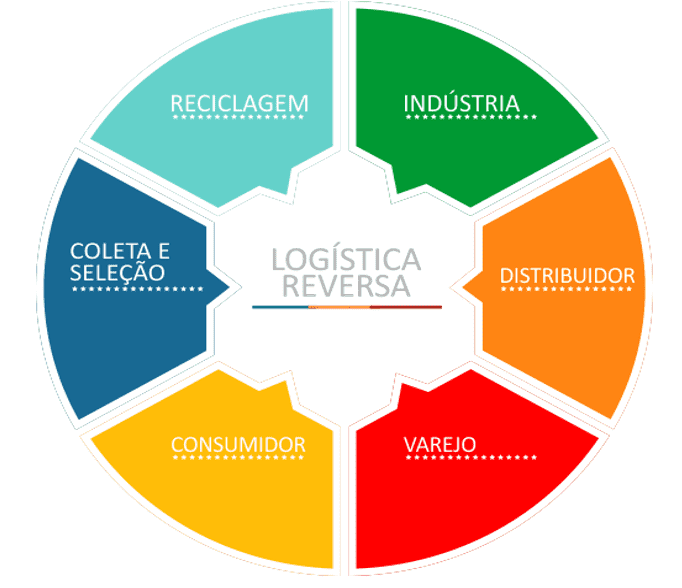
Source: http://jplogistica.com.br/
Decree No. 9,177 of 10/23/2017 in Article 1 establishes standards to ensure isonomy in the supervision and fulfillment of obligations attributed to manufacturers, importers, distributors and traders of products, their waste and their packaging subject to mandatory reverse logistics.
Thus, the obligations and responsibilities, from the production and marketing of products and other solid waste generated becomes a shared obligation by the various users of these products and waste generated, whether manufacturers, importers, distributors, traders and or consumers, thus ensuring the reuse, collection and conscious disposal of these materials, giving the appropriate purpose, whether disposal or reuse of the generated waste.
It is worth mentioning that in the case of pharmaceutical or medical products, the Health Services Waste Management Plan (PGRSS) requires that improper disposal of medicines may lead to administrative penalties, according to Federal Law No. 12,305, of 08/02/2010.
Figure 4. MP clears irregular disposal

Source: tribunadonorte.com.br
FINAL CONSIDERATIONS
Given the present work, and considering the great importance with regard to the results of logistics management of supplements and finished products, focused on the area of medicines and correlated to serve the public sector, it is concluded that the investment in the human factor, as well as in the awareness of people using these products, by the public sector, may provide a more modern and broad view in the logistics management of these products and waste generated , considering that the involvement of all those responsible in the logistics process of materials and medicines and their correqueres, may culminate in specific results, aiming at greater agility in the processes involved with the return and proper destination of these products or insums generated by the drug chain. This ends up avoiding the waste of public resources and poor performance of the demanding sector, thus ensuring the effective performance of the reverse logistics cycle of medicines, which will certainly generate the control of the garbage generated daily by the increasing consumption of these products by the requesting public. In turn, nature conservation, which is so necessary and indispensable for life on this planet, will also be guaranteed.
REFERENCES
A CONFUSÃO DA LOGÍSTICA REVERSA DE MEDICAMENTO NO BRASIL; Viviane Massi, ICTQ, 2020; Disponível em: https://www.ictq.com.br/varejo-farmaceutico/844-a-confusao-da-logistica-reversa-de-medicamentos-no-brasil
AGÊNCIA BRASIL, 2011. Empresa Brasil de Telecomunicação. Relatório de ONU Aponta Consumo Abusivo de Remédios no Brasil. Disponível em:< http://agenciabrasil.ebc.com.br/noticia/2011-06-23/relatorio-da-onu-aponta-consumo-abusivode-remedios-no-brasil>. Acesso em 04 abril 2012.
ANVISA, 2010. Agência Nacional de Vigilância Sanitária. Diagnóstico situacional da promoção de medicamentos em unidades de saúde do SUS.
BARBOSA, M. O e-comerce é um parceiro estratégico para o setor da saúde. Revista Diagnostico, São Paulo, V. 4, n. 17, p. 48, Set. /Out. 2012. Disponível em: < http://www.diagnosticoweb.com.br/noticias/gestao/direto-ao-ponto-o-e-commerce-e-parceiro-estrategico-para-asaude.html> Acesso em: 01/06/2017.
BRASIL. Ministério da Saúde. Apoio à Gestão Hospitalar: E-SUS Hospitalar. Disponível em: http://portalsaude.saude.gov.br/index.php/oministerio/principal/secretarias/sgep/sgepnoticias/23553-apoio-a-gestao-hospitalar-e-sus-hospitalar.Acessado em: 10/10/2017
BRASIL. Ministério do Meio Ambiente. Conselho Nacional do Meio Ambiente (CONAMA). Resolução no 358, de 29 de abril de 2005. Dispõe sobre o tratamento e a disposição final dos resíduos dos serviços de saúde e dá outras providências.
BRASIL. Portaria MS n. 3.916, de 30 de outubro de 1998. Política Nacional de Medicamentos. Brasília: Ministério da Saúde, 1998.
BRASIL. Resolução ANVISA RDC n. 306, de 7 de dezembro de 2004. Dispõe sobre o Regulamento Técnico para o gerenciamento de resíduos de serviços de saúde. Brasília: Ministério da Saúde, 2004.
BRASIL. Resolução CONAMA n. 358, de 29 de abril de 2005. Dispõe sobre o tratamento e a disposição final dos resíduos de serviços de saúde e dá outras providências. Brasília: Ministério do Meio Ambiente, 2005.
BUENO, CS., WEBER, D., OLIVEIRA, KR. Farmácia caseira e descarte de medicamentos no bairro Luiz Fogliatto do município Ijuí-RS. Rev. Ciências Farm. Básica Apl. 2009; 30(2):75-82.
DEMAJOROVIC, J.; et. al. Logística reversa: como as empresas comunicam o descarte de baterias e celulares? Revista de Administração de Empresas, v. 52, n. 2, p.165-178, 2012.
EICKHOFF, P.; HEINECK, I.; SEIXAS, L. J. Gerenciamento e destinação final de medicamentos: uma discussão sobre o problema. Revista Brasileira de Farmácia, 90 (1), 64-68, 2009.
FALQUETO, E; KLIGERMAN, D. C; ASSUMPÇÃO, R. F. Como realizar o correto descarte de resíduos de medicamentos? Ciência & Saúde Coletiva, v.15, n.2, p. 3283-3293, 2010.
FALQUETO, E; KLIGERMAN, D. C. Diretrizes para um Programa de Recolhimento de Medicamentos vencidos no Brasil. Ciência & Saúde Coletiva, v. 18, n. 3, p. 883-892, 2013.
GOUVEIA, N. Resíduos sólidos urbanos: impactos socioambientais e perspective de manejo sustentável com inclusão social. Rev. Ciência & Saúde Coletiva, 17 (6), 1503-1510, 2012.
_______. MINISTÉRIO DA SAÚDE. Apoio à Gestão Hospitalar: E-SUS Hospitalar. Disponível em: http://portalsaude.saude.gov.br/index.php/oministerio/principal/secretarias/sgep/sgepnoticias/23553-apoio-a-gestao-hospitalar-e-sus-hospitalar.
[1] Graduated in Pharmacy.
Sent: March, 2020.
Approved: November, 2020.















