ORIGINAL ARTICLE
RIBEIRO, Rachel Melo [1], SILVA, Gizele de Oliveira Santos [2], MAIA, Ellen Julli da Silva Passos [3], PEREIRA, Simone Bom Jardim [4], COSTA, Izolda Souza [5], CAMPOS, Matheus Brandão [6], SILVA JUNIOR, Jose Ribamar da [7], PINHEIRO NETO, Vicente Ferrer [8], CALDAS, Germana Freire Rocha [9], CARTAGENES, Maria do Socorro de Sousa [10], BORGES, Marilene Oliveira da Rocha [11], BORGES, Antonio Carlos Romão [12]
RIBEIRO, Rachel Melo. et al. Cardioprotective profiling of syzygium cumini: evidence in experimental model of ischemic injury. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year. 08, Ed. 07, Vol. 01, pp. 158-176. April 2023. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/health/cardioprotective-profiling, DOI: 10.32749/nucleodoconhecimento.com.br/health/cardioprotective-profiling
ABSTRACT
Syzygium cumini is a medicinal plant traditionally used to treat cardiometabolic disorders. This study proposed to evaluate the cardiopreventive effect of the hydroalcoholic extract from the S. cumini leaves (HESc) on ischemic injury isoproterenol (ISO)-induced in rats. Male albino Wistar rats (Rattus norvegicus) were pre-treated with HESc or water for 15 days. They received a challenge with ISO (85 mg/kg) on the 14th and 15th days for induction of ischemic injury. For all animals were obtained electrocardiographic parameters predictive of the lesion in addition to evaluation of troponin I changes in animals. We also realized a phytochemical screening, and in vitro antioxidant activity including 1,1-Diphenyl-2-picrylhydrazyl radical. HESc was found to be rich in flavonoids, and high antioxidant potential. Addictionaly, increased the survival rate and reversed ventricular remodeling caused by isoproterenol-induced ischemic injury in a dose-dependent manner. Inhibition of ST segment elevation, impairmentf QT interval prolongation, reduction of Troponin I levels also were observed in animals pretreated with HESc. Our results suggest that pretreatment with HESc reduces myocardial injury and simple phenols and flavonols are likely to be responsible for its effect on cardiomyocyte integrity, confirming that S. cumini is a promising plant source of bioactive compounds for the treatment of ischemic syndromes.
Keywords: Medicinal Plant, Bioprospecting, Natural products, Flavonols, Plant Extract.
BACKGROUND
Syzygium cumini (L.) Skeels (Myrtaceae), popularly known in Brazil as Jambolão, is extensively used to treat obesity, diabetes, and hypertension (Migliato et al., 2006). Studies indicate the presence of myricetin, myricitrin, quercetin, kaempferol, and simple phenols such as ellagic acid, ferulic acid and gallic acid (Ruan; Zhang; Lin, 2008; Ayyanar and Subash-Babu, 2012; Sanches et al., 2016).
- cumini possesses antihyperglycemic (Villasenor and Lamadrid, 2006; Sharma et al., 2011), antioxidant, antiatherosclerotic and antihyperlipidemic properties (Ruan; Zhang; Lin, 2008; Tanwar et al., 2011; Sharma et al., 2012), and cardioprotective activity reported for the fruits (Shukla et al., 2014) and seeds (Mastan et al., 2009; Atale et al., 2017).
In recent years, our laboratory has been conducting toxicological and pharmacological tests in animals with a standardized hydroalcoholic extract of S. cumini leaves (HESc). The toxicological analysis in rodents revealed that the extract did not exhibit toxicity (Silva et al., 2012). In the pharmacological tests, we demonstrate a hypotensive, antihypertensive, vasorelaxant and antispasmodic effect, and a voltage-dependent block of calcium channel current (Ribeiro, 2007; Monteiro et al., 2018).
In this study, we evaluated the preventive effect of HESc on IHD in an experimental model of isoproterenol-induced myocardial ischemia in rats. The findings of HESc on electrocardiography, estimation of cardiac hypertrophy, and biochemical analyze of troponin I of the animals contribute to the scientific validation of S. cumini leaves as complementary or alternative therapy in the treatment or prevention of ischemic cardiovascular disorders.
METHODS
PLANT MATERIAL AND PREPARATION OF EXTRACT
Leaves were collected as previously described by Ribeiro et al. (2018), and a voucher specimen was deposited under No. 1069 (herbarium of the ‘Prof. Dr. Berta Lange de Morretes “Medicinal Plant Garden, UFMA). The leaves were dried at room temperature, followed by maceration of the leaf powder (300 g) in 70% ethanol (1:3 w/v), concentration in a rotary evaporator under reduced pressure at a temperature below 60ºC and lyophilized, with a dry weight of 49.8 g and yield 16.6% (Ribeiro et al., 2018).
DETERMINATION OF TOTAL PHENOLIC CONTENT (TPC) AND TOTAL FLAVONOID CONCENTRATION (TFC)
The TPC of all samples was determined with the Folin−Ciocalteu reagent and 20% sodium carbonate, method described by Dutra et al. (2014). Gallic acid (GA) was used as standard, and results expressed as GA equivalents per 100 g dry extract (GAE/100 g). In addition, the TFC was determined by the aluminium chloride colorimetric method described by Dutra et al. (2008). Quercetin (QUE) was used as standard, and results expressed as QUE equivalents per 100 g dry extract (QUE/100 g). All experiments were done in triplicate.
DETERMINATION OF DPPH•SCAVENGING ASSAY
The antioxidant activity of the extracts was measured based on the scavenging activity of the stable 1, 1- diphenyl 2-picrylhyorazyl (DPPH) free radical according to the method described by Brand-Williams, Cuvelier and Berset et al. (1995) with slight modifications. Gallic acid was used as a positive control. DPPH free radical scavenging ability (%) was calculated by using the:
DPPH scavenging activity (%) = 100 – [(A sample –A blank) x 100/ A control]
where Asample = absorbance of the sample after 30 min of reaction, Ablank = absorbance of the blank, and Acontrol = absorbance of the control.
EXPERIMENTAL ANIMALS
Male 08-week-old Wistar rats (Rattus norvegicus), weighing 200 to 250 g, obtained from the Animal House of UFMA were used. The animals were housed under controlled conditions of temperature (21±2 ºC) under a 12 h light-dark cycle and humidity from 55 – 65%. The experimental protocols were submitted to and approved by the Animal Research Ethics Committee from State University of Maranhão, Brazil, under license number 23115.013098/2020-73.
EXPERIMENTAL PROTOCOLS
Before starting the experiments, the animals were kept in the laboratory of Pharmacology of the UFMA by a period of one week for adaptation and monitored before and during the experimentation. After, the animals were randomly divided into the different groups and were treated by gavage (oral administration, gavage) with water (controls) or HESc dissolved in water, receiving 0.1 mL/100 g (rats).
Forty Wistar rats were divided into four groups of 10 animals each: healthy Control0.1 ml water/100 g), ISO (0.1 ml water/100 g + ISO), high-dose HESc (500 mg/kg/day + ISO), and low-dose HESc (250 mg/kg/day + ISO). The animals were pretreated with HESc for 15 consecutive days. Acute myocardial infarction was induced on days 14 and 15 by subcutaneous injection of ISO diluted in 2 mL saline (0.9% NaCl; 85 mg/kg) (Rajadurai and Prince, 2007; Hassan et al., 2015), in all groups, except for the healthy Control group that received only saline by the same route. Clinical signs of toxicity or mortality were recorded daily. At the end of the experiment (day 16), the animals were anesthetized (50 mg/kg ketamine and 10 mg/kg xylazine) for the subsequent experimental procedures.
The doses for HESc evaluated in this study were based on previous results obtained for Wistar in toxicity assay (Silva et al., 2012), and spontaneously hypertensive (SHR) rats, whose dose 500 mg/kg/day was able to promote changes in the animals’ heart rate (Ribeiro et al., 2014). Furthermore, this dose promoted a reduction in the weight of the left and right ventricles in hypertensive animals treated for 08 weeks (unpublished data), which motivated us to evaluate it in a shorter treatment time (15 days), together with a lower dose in cardiac ischemic injury model, to establish a time-dependent and dose-dependent effect relationship, respectively.
Electrocardiography
Electrocardiograms (ECG) were obtained from the anesthetized animals using a veterinary electrocardiograph (DL 600, Delta Life®, São Paulo, Brazil). The QTc interval was calculated using Bazzet’s equation: QTc= QTinterval/√R-R interval.
Estimation of cardiac hypertrophy and Biochemical analyze.
The heart was removed, and washed with ice-cold saline, weighed to obtain the heart weight-to-body weight ratio adapted by Yousefi et al. (2014). The heart was then cut to remove the left ventricle (LV) and the LV weight-to-body weight ratio was calculated to assess the degree of congestion.
After ECG, blood was collected from the abdominal artery of anesthetized rats into tubes without anticoagulant and centrifuged at 3,500 rpm for 10 min. Serum was separated, and Troponin I was measured by a rapid immunochromatographic assay (WAMA Diagnóstica®).
STATISTICAL ANALYSIS
The results were expressed as the mean ± standard error of the mean (S.E.M.). One-way analysis of variance (ANOVA) followed by the Tukey post-test was used for multiple comparisons and the Student t-test for comparison of unpaired data. A p value <0.05 was considered significant. Statistical analysis was performed using the GraphPad Prism® 9.0 program.
RESULTS
TPC AND TFC OF HESC
The content of total phenols and flavonoids in HESc was 300.7 ± 3.9 GAE/100 g dry weight of extract and 83.5 ± 2.4 QUE/100 g dry weight of extract, respectively. These results suggest a high content of phenolic compounds, particularly flavonoids, in leaves of S. cumini.
DPPH RADICAL SCAVENGING ACTIVITY OF HESC
HESc showed the highest DPPH scavenging activity, at 91.73 ± 0.30 % with an IC50 value of 4.02 ± 0.13 μg/mL. IC50 value of the positive control Gallic Acid was 1.55 ± 0.44 μg/mL (92.30 ± 0.30 %).
HESC ENHANCES THE SURVIVAL OF RATS WITH ISO-INDUCED ISCHEMIC INJURY
The survival rate of animals that received the low-dose (250 mg/kg/day) and water (ISO group) was 72.72% and 63.64%, respectively (Fig. 1), and with the highest dose of HESc (500 mg/kg/day) was 100%. These results suggest that a HESc dose of 500 mg/kg/day may effectively prevent death and increase the chance of survival of animals with ischemic injury.
Fig 1. Syzygium cumini leaves increases chances of survival after ISO-induced ischemic injury
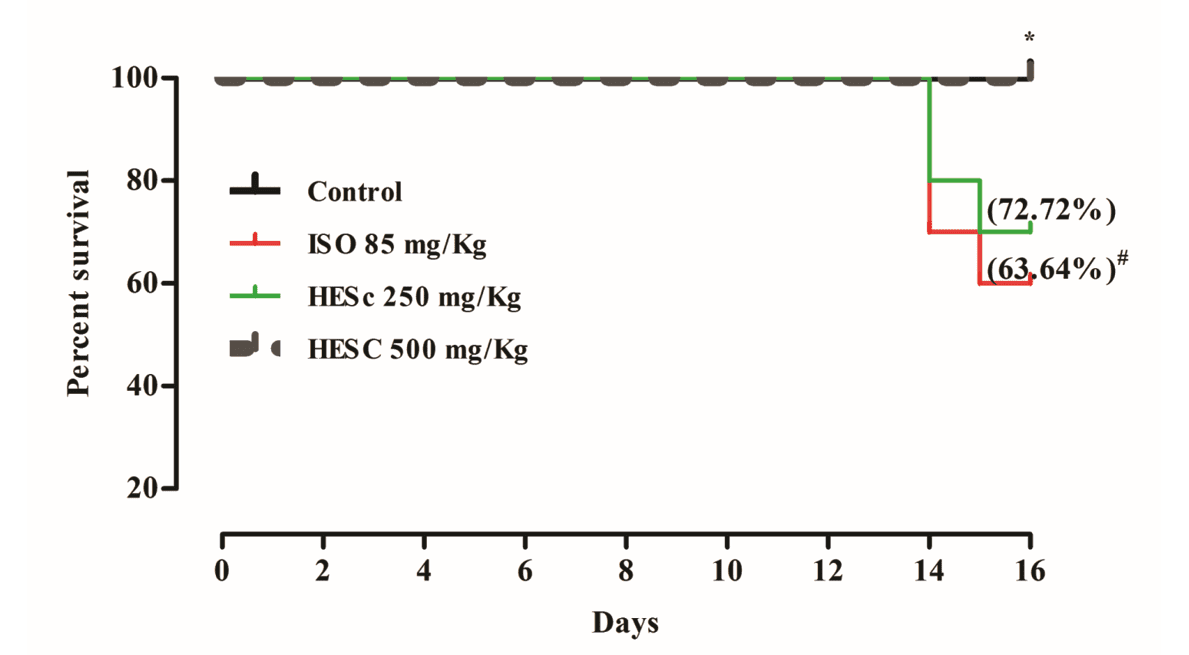
The survival rate of ISO-injected animal treated with Water and 250 mg/kg/d and 500 mg/kg/day of HESc. #p< 0.05 vs. Control group; *p < 0.05 vs. Water + ISO.
EFFECT OF HESC ON ELECTROCARDIOGRAPHIC PATTERN AND PARAMETERS
The data regarding ST and PR segments, QRS complex, QT and QTc intervals and R-R interval are shown in Figs. 2 and 3. The ECG recording of healthy control animals indicates a regular and uniform sinus rhythm (Fig. 3). ISO-induced animals exhibited significant (p<0.001) ST segment elevation when compared to the Control group (Figs. 2A and 2B). Pretreatment with HESc (500 mg/kg/day) caused significant (p<0.001) cardioprotection, with a more than 50% decline in ST segment elevation compared to the ISO-induced group and values like the Control group (Fig. 2A).
Fig 2. HESc prevents ISO-induced cardiac electrical changes of animals after ISO-induced myocardial injury
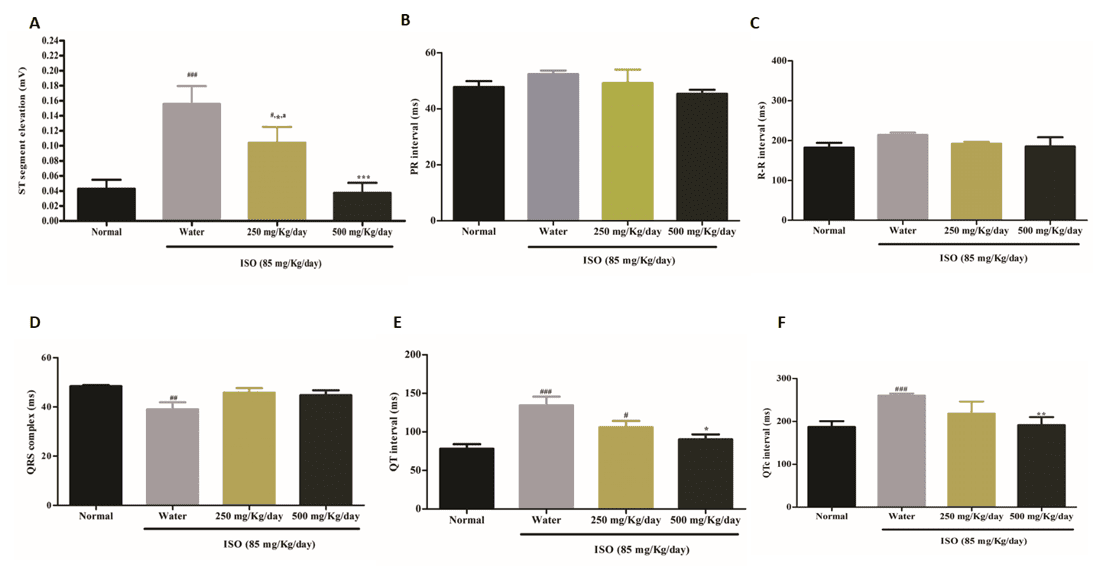
(A) ST segment. (B) PR segment. (C) R-R interval. (D) QRS complex. (E) QT interval. (F) QTc interval. Results are expressed as mean ± S.E.M. (n = 6-10). ###p <0.001 vs Control group; ##p <0.01 vs Control group; ***p <0.001 vs Water + ISO; ap <0.05 vs HESc 500 mg/kg + ISO (ANOVA, Tukey).
In the ISO-induced group, a significant decline (p<0.01) in the QRS (Fig. 2D) complex and an increase in the QT (p<0.001) and QTc intervals (p<0.001) were observed when compared to the control group (Figs 2E, 2F and 3B) with no significant alterations in the PR or R-R interval (Figs 2B and 2C). Pretreatment with HESc (500 mg/kg/day) resulted in significant cardioprotection, with restoration of the QT and QTc intervals when compared to the ISO-induced group (p<0.05 and p<0.01, respectively). This effect was more pronounced when compared to the lower dose, thus suggesting a dose-dependent cardioprotective effect of S. cumini leaves (Figs 3C and 3D).
Fig 3. Representative image of the electrocardiogram of animals after ISO-induced myocardial injury
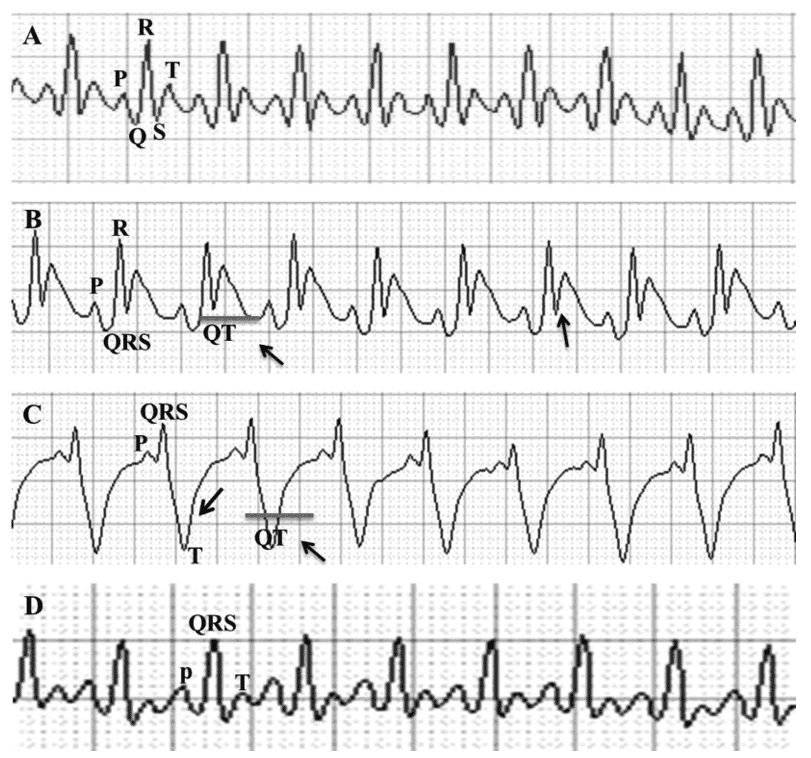
(A) Normal control group showing with electrocardiographic reccording without changes. (B) Water + ISO showing ST segment elevation and QT interval prolongation. (C) HESc 250 mg/Kg/day + ISO group, presenting myocardial ischemia and QT interval prolongation. (D) HESc 500 mg/Kg/day + ISO, presenting a sinus rhythm with a slight increase in amplitude of the P wave.
EFFECT OF HESC ON THE ESTIMATION OF HYPERTROPHY AND TROPONIN I
A significant increase was observed in the heart weight/body weight ratio (Fig. 4A) and LV weight/body weight ratio (Fig.4B) in animals of the ISO-induced group. Pretreatment with the
highest dose of HESc for 15 days reduced the heart weight (p<0.001) and LV weight ratios (p<0.001) when compared to ISO-induced animals. In contrast, the lower dose reduced the LV weight/body weight ratio (p<0.05), but the ratio differed from the Normal Control group (p<0.05).
Fig 4. HESc reduced ISO-induced ventricular hypertrophy
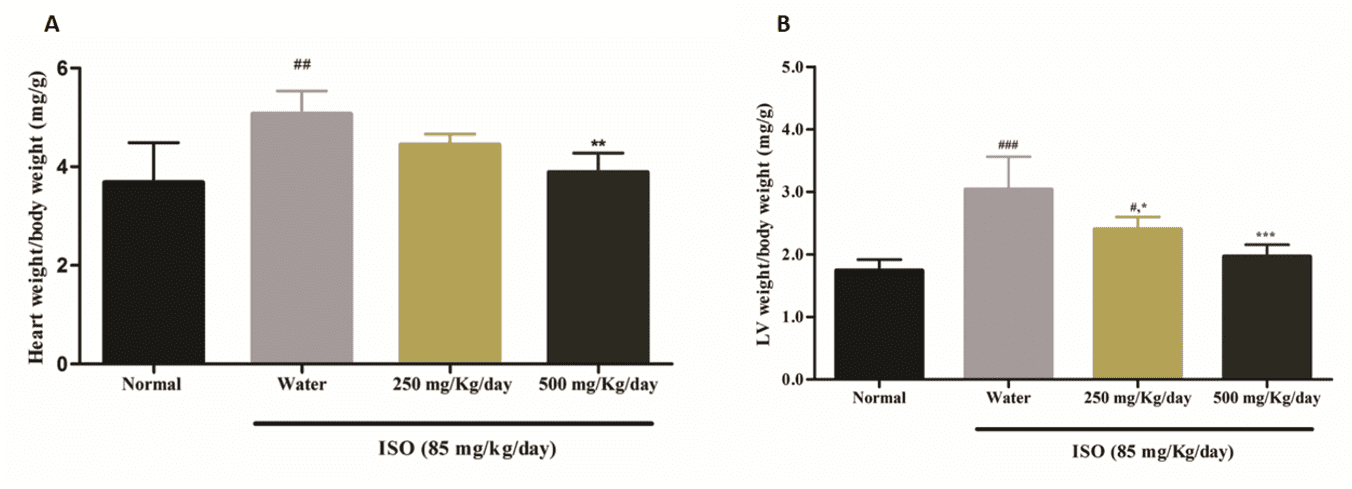
(A) Heart weight/body weight (mg/g). (B) LV weight/body weight (mg/g). All values are indicated as mean ± S.E.M. (n=5). ###p < 0,0001 vs. Normal Control, ***p <0,001 vs. Water + ISO, ****p <0,0001 vs. Water + ISO (ANOVA, Tukey).
Troponin I was positive in all animals of the ISO-induced heart injury group and absent in serum of all animals pretreated with the high dose of HESc (500 mg/kg/day) or Control group (Table 1).
Table 1. Effect of Syzygium cumini on serum troponin I levels measured by immunochromatographic assay in animals submitted to acute myocardial infarction
|
Animal |
Groups | |||
| Normal Control | Water + ISO | HESc 250 mg/Kg + ISO | HESc 500 mg/Kg + ISO | |
| 1 | n | p | p | n |
| 2 | n | p | p | n |
| 3 | n | p | p | n |
| 4 | n | p | n | p |
| 5 | n | p | p | n |
Seurce: Elaborated by the authors.
n: absence of troponin I; p: presence of troponin I. Troponin I was measured with the WAMA Diagnóstica® rapid immunochromatographic assay (n = 5).
DISCUSSION
This is the first study that evidences the therapeutic cardiopreventive potential of Syzygium cumini leaves, observed in ECG analysis or normalization of Troponin I, and probably is attributed to the high content of phenolic and flavonols compounds, and include the simple preparation of the extract and short treatment time.
One of the mechanisms supposedly involved in irreversible ISO-induced myocardial ischemic injury is calcium overload (Yousefi et al., 2014). Responses include an increase in cardiac contractility and pathological alterations such as hypertrophy and cell apoptosis (Yousefi et al., 2014; Bloom and Davis, 1972). The main route of entry of calcium into cardiomyocytes is through L-type calcium channels. In this respect, the blockage of these channels has been suggested to play a preventive role in ischemic injury. Previous results obtained by our group for HESc have shown a possible effect of the leaf extract of S. cumini on the inhibition of L-type calcium channels (Ribeiro et al., 2018), in agreement with the present study in which HESc prevented ventricular remodeling in a dose-dependent manner.
IHD in general are associated with changes in the electric properties of the heart and ECG is used as a diagnostic tool of cardiomyopathies (Patel et al., 2010). ST segment elevation is a sensitive marker of acute myocardial infarction and is related to the difference in membrane potential between ischemic and non-ischemic areas, with a consequent loss of cell membrane function ((Patel et al., 2010; Zhou et al. 2008). ST segment elevation has been observed in patients with myocardial ischemia (Peacock et al., 2007) and in rats with ISO-induced myocardial injury (Kela; Reddy; Thombre, 1980; Rajadurai and Prince, 2007). Some authors suggest ST segment abnormalities to be an indicator of morbidity and mortality due to ischemic heart disease (Achari et al., 2008; Mozos and Caraba, 2015). Our results showed that ISO-induced ischemic injury promoted changes in the ECG recording and ST segment elevation was suppressed by pretreatment with the highest dose of HESc.
Other ECG alterations are expected for the experimental model used, such as a reduction of the R-R interval and prolongation of the QT interval (Mikušová et al., 2009). No differences in the R-R interval were observed in the present study. These intervals remained elevated in the groups even after the administration of ISO, suggesting bradycardia of the animals. This fact might be explained using ketamine and xylazine as anesthetic combination, which is known to cause bradycardia in rats (Sano et al., 2016).
Prolongation of the QT interval is commonly associated with cardiac hypertrophy, more specifically LV hypertrophy, whose physiopathological relevance involves the development of mechanical dysfunction, pump failure and sudden death (Jose and Gupta, 2004; Davey et al., 1994). In the present study, HESc restored the duration of the QT interval to normal levels, corroborating the dose-dependent effect of S. cumini leaves on cardiac remodeling. Additionally, the Troponin I biomarker evaluated in this study is used as a laboratory tool for diagnosis of acute myocardial infarction in humans (Kim et al., 2014; Fathil et al., 2015). The results showed a dose-dependent effect for HESc to prevent the plasma elevation of troponin I.
With respect to chemical composition, our results indicate that HESc presented higher content of polyphenols compared to leaf extract obtained by (Sanches et al., 2016), which identified the presence of simple phenols like gallic acid and myricetin-derived flavonols in leaf extract. Ryu et al. (2016) showed that gallic acid prevents LV hypertrophy and fibrosis ISO-induced in mice. Zern et al. (2005) observed that supplementation of patients with an extract containing myricetin confers cardioprotection against oxidative damage after consumption for 30 days. This finding was corroborated by Tiwari et al. (2009) who demonstrated the protective role of myricetin from Vitis vinifera in reducing the ISO-induced cardiac toxicity in rats. We may therefore suggest that the cardiopreventive activity of HESc against ischemic injury observed in this study is related in part to the presence of these compounds (Figure 5).
Detection of the antioxidant potential of plant extracts can be evaluated by a relatively simple spectrophotometric method through the DPPH assay. This method consists in evaluating the antioxidant capacity through the sequestering activity of the free radical 2,2-diphenyl-1-picryl-hydrazine-DPPH. The DPPH radical by the action of an antioxidant is reduced to form diphenyl-picryl hydrazine, yellow staining (Rao et al., 2010; Lee et al., 2007).
The results indicate a high percentage of inhibition for HESc positively correlated with phenolic and total flavonoid content. The antioxidant potential of S. cumini leaves was evidenced in other studies that corroborate our results (Ruan; Zhang; Lin, 2008; Sanches et al., 2016; Eshwarappa et al., 2014).
Fig 5. Proposed for the Cardiopreventive Action of Syzygium cumini
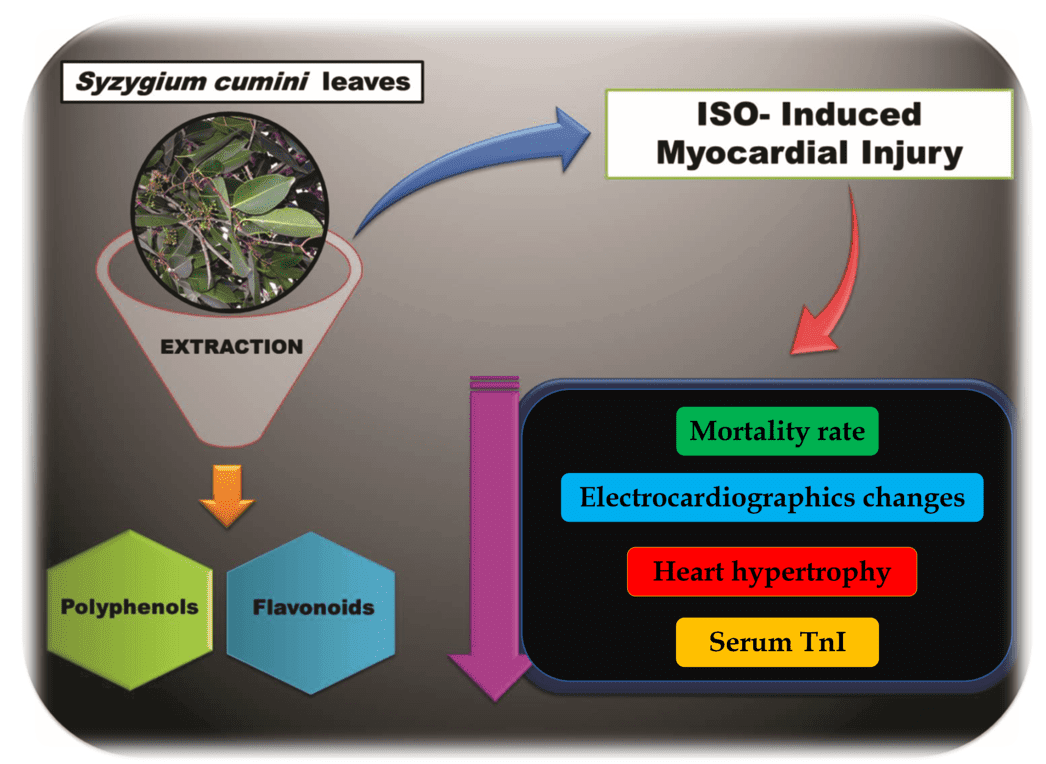
The large amount of polyphenols and the high content of flavonoids contribute to the high antioxidant potential of the plant extract, whose previous oral treatment for 15 days was able to reduce mortality, reverse electrical alterations, prevent cardiac hypertrophy, suppress heart failure, and elevation of TnI levels.
CONCLUSIONS
In conclusion, the results characterize a cardioprotective effect of S. cumini leaves in animals with ischemia by ISO. Further studies are needed to elucidate the mechanism underlying the preventive effect of S. cumini on the myocardial ischemic injury. Thus, it is hoped to contribute to the scientific validation of this species as a promising plant source of bioactive compounds in the treatment of ischemic heart diseases.
COMPETING INTERESTS
The authors declare that they have no competing interests.
Funding:
This work was supported by Fundação de Amparo à Pesquisa do Estado do Maranhão, Brasil (FAPEMA) project 2012–2014 (Reference nº: APP-UNIVERSAL-00354/12) and 2014-2016 (Reference nº: UNIVERSAL-00788/14). This study was conducted at the Federal University of Maranhão (UFMA) in cooperation with CEUMA University and State University of Maranhão.
ACKNOWLEDGEMENTS
This study it was conducted in cooperation with Universidade Federal do Maranhão (UFMA), Instituto Federal do Maranhão (IFMA) and Universidade Estadual do Maranhão (UEMA).
REFERENCES
ACHARI, V. et al. Short-term mortality and complications in ST elevation myocardial infarction–the Heart Hospital experience. J Indian Med Assoc. 2008; 106: 650-4.
ATALE, N. et al. Synthesis and Characterization of Sygyzium cumini Nanoparticles for Its Protective Potential in High Glucose-Induced Cardiac Stress: a Green Approach. Appl Biochem Biotechnol. 2017;18:1140-54.
AYYANAR, M.; SUBASH-BABU, P. Syzygium cumini (L.) Skeels: A review of its phytochemical constituents and traditional uses. Asian Pac J Trop Biomed. 2012;2: 240-6.
BLOOM, S.; DAVIS, D. L. Calcium as Mediator of Isoproterenol-Induced Myocardial Necrosis. The American Journal of Pathology. 1972;69: 459-70.
BRAND-WILLIAMS, W.; CUVELIER, M. E.; BERSET, C. Use of free radical method to evaluate antioxidant activity. Lebensm. Wiss. Technol. 1995;28: 25-30.
DAVEY, P. P. et al. QT interval dispersion in chronic heart failure and left ventricular hypertrophy: relation to autonomic nervous and Holter tape abnormalities. Br Heart J. 1994; 71: 268-73.
DUTRA, R. P. et al. Avaliação farmacognóstica de geoprópolis de Melipona fasciculata Smith da Baixada maranhense, Brasil. Braz J. Pharmacogn. 2008;18: 557-62.
DUTRA, R. P. et al. Phenolic Acids, Hydrolyzable Tannins, and Antioxidant Activity of Geopropolis from the Stingless Bee Melipona fasciculata Smith. J Agric Food Chem. 2014;62: 2549-57.
ESHWARAPPA, R. S. B. et al. Antioxidant activity of Syzygium cumini leaf gall extracts. BioImpacts. 2014;4:101–7.
FATHIL, M. F. et al. Diagnostics on acute myocardial infarction: Cardiac troponin biomarkers. Biosens Bioelectron. 2015;70: 209-20.
HASSAN, M. Q. et al. Benidipine prevents oxidative stress, inflammatory changes and apoptosis related myofibril damage in isoproterenol-induced myocardial infarction in rats. Toxicol Mech Methods. 2015; 25: 26-33.
JOSE V. J.; GUPTA, S. N. Mortality and morbidity of acute ST segment elevation myocardial infarction in the current era. Indian Heart J. 2004; 56: 210-4.
KELA, A. K.; REDDY, P. L.; THOMBRE, D. P. ECG findings in normal rats and after administration of isoproterenol. Ind J Physiol Pharmacol. 1980; 24: 84–90.
KIM, T. K. et al. Point-of-Care Fluorescence Immunoassay for Cardiac Panel Biomarkers. J Clin Lab Anal. 2014;28: 419-27.
LEE, Y. R. et al. Antioxidant activities of ethanol extracts from germinated specialty rough Rice. Food Sci Biotechnol. 2007;16:765-70.
MASTAN, S. K. et al. Cardioprotective effect of methanolic extract of Syzygium cumini seeds on isoproterenol-induced myocardial infarction in rats. Pharm Lett. 2009; 1: 143-9.
MIGLIATO, K. F. et al. Ação farmacológica de Syzygium cumini (L.) Skeels. Acta Farm. Bonaerens. 2006; 25:310-4.
MIKUŠOVÁ, A. et al. Myocardial remodelling induced by repeated low doses of isoproterenol. Can J Physiol Pharmacol. 2009; 87: 641-51.
MONTEIRO, F. S. et al. Antidiarrhoeal and antispasmodic activity of leaves of Syzygium cumini L. (Myrtaceae) mediated through calcium channel blockage. Afr. J. Pharm. Pharmacol. 2018; 12:11-18.
MOZOS, I.; CARABA, A. Electrocardiographic Predictors of Cardiovascular Mortality. Dis Markers. 2015;2015:727401.
PATEL, V. et al. Cardioprotective effect of melatonin against isoproterenol induced myocardial infarction in rats: a biochemical, electrocardiographic and histoarchitectural evaluation. Eur J Pharmacol. 2010; 644: 160-8.
PEACOCK W. F. et al. Reperfusion strategies in the emergency treatment of ST-segment elevation myocardial infarction. Am J Emerg Med. 2007; 25: 353-66.
RAJADURAI, M.; PRINCE, P. S. M. Preventive effect of naringin on cardiac markers, electrocardiographic patterns and lysosomal hydrolases in normal and isoproterenol-induced myocardial infarction in Wistar rats. Toxicology. 2007; 230: 178–88.
RAO, A. S. et al. The antioxidant and antiproliferative activities of methanolic extracts from Njavara rice bran. BMC Complement Altern Med. 2010;10: 1-9.
RIBEIRO R. M. Estudo da Atividade Hipotensora das Folhas de Syzygiumj jambolanum DC (Jambolão). MSc. Thesis, Universidade Federal do Maranhão. 2007 Available in: <https://tedebc.ufma.br/jspui/handle/tede/1055>.
RIBEIRO R. M. et al. Antihypertensive Effect of Syzygium cumini in Spontaneously Hypertensive Rats. Evid Based Complement Alternat Med. v. 2014, pp. 1-7. 2014.
RIBEIRO, R. M. et al. Extract and chloroform fraction from Syzygium cumini leaves with vasorelaxant effect mediated by inhibition of calcium channels. Afr. J. Pharm. Pharmacol. v. 12, pp. 151-158. 2018.
RUAN, Z. P; ZHANG, L. L; LIN Y. M. Evaluation of the antioxidant activity of Syzygium cumini leaves. Molecules. v. 13, pp. 2545-2556. 2008.
RYU, Y. et al. Gallic acid prevents isoproterenol-induced cardiac hypertrophy and fibrosis through regulation of JNK2 signaling and Smad3 binding activity. Sci Rep. v. 6, pp. 1-14. 2016.
SANO, Y. et al. Effects of various types of anesthesia on hemodynamics, cardiac function, and glucose and lipid metabolism in rats. Am J Physiol Heart Circ. v. 311, H1360-H1366. 2016.
SANCHES, J. R. et al. Polyphenol-Rich Extract of Syzygium cumini Leaf Dually Improves Peripheral Insulin Sensitivity and Pancreatic Islet Function in Monosodium L-Glutamate-Induced Obese Rats. Front Pharmacol. V. 7, pp. 1-16. 2016.
SHARMA, S. B. et al. Ameliorative Effect of Active Principle Isolated fromSeeds of Eugenia jambolana on Carbohydrate Metabolism in Experimental Diabetes. Evid Based Complement Alternat Med. v. 2011, 1-9. 2011.
SHARMA, A. K. et al. Syzygium cumini ameliorates insulin resistance and beta-cell dysfunction via modulation of PPAR, dyslipidemia, oxidative stress, and TNF-alpha in type 2 diabetic rats. J. Pharmacol. Sci. v. 119, pp. 205–213. 2012.
SHUKLA, S. K. et al. Eugenia jambolana pretreatment prevents isoproterenol-induced myocardial damage in rats: evidence from biochemical, molecular, and histopathological studies. J Med Food. V. 17, pp. 244–253. 2014.
SILVA, S. N. et al. The toxicity evaluation of Syzygium cumini leaves in rodents. Rev Bras Farmacogn. v. 22. pp. 102–108. 2012.
TANWAR, R. S. et al. Antiatherosclerotic potential of active principle isolated from Eugenia jambolana in streptozotocin-induced diabetic rats. Evid Based Complement Alternat Med. v. 2011, pp. 1-9. 2011.
TIWARI, R. et al. Cardioprotective potential of myricetin in isoproterenol-induced myocardial infarction in Wistar rats. Phytother Res. v. 23, pp. 1361-1366. 2009.
VILLASENOR, I. M.; LAMADRID, M. R. Comparative anti-hyperglycemic potencials of medicinal plants. J Ethnopharmacol. v. 104, 129-131. 2006.
YOUSEFI, K. et al. Marrubium vulgare L. methanolic extract inhibits inflammatory response and prevents cardiomyocyte fibrosis in isoproterenol-induced acute myocardial infarction in rats. BioImpacts. v. 4, 21-27. 2014.
ZERN, T. L. et al. Grape polyphenols exert a cardioprotective effect in pre- and postmenopausal women by lowering plasma lipids and reducing oxidative stress. J Nutr. v. 135, pp. 1911-1917. 2005.
ZHOU, R. et al. Cardioprotective effect of fluvastatin on isoproterenol-induced myocardial infarction in rat. Eur J Pharmacol. v. 586, pp. 244-250, 2008.
[1] Advisor. PhD in Biotechnology. ORCID: https://orcid.org/0000-0003-3684-3061. Currículo Lattes: http://lattes.cnpq.br/4752952470368965.
[2] Medical School (UFMA). ORCID: https://orcid.org/0000-0002-0150-7212. Currículo Lattes: http://lattes.cnpq.br/3679081425109665.
[3] Graduation Biochemistry Pharmacy. ORCID: https://orcid.org/0009-0009-8092-5647. Currículo Lattes: http://lattes.cnpq.br/3679081425109665.
[4] Graduation Biochemistry Pharmacy. ORCID: https://orcid.org/0009-0006-6760-5743. Currículo Lattes: https://lattes.cnpq.br/2927956835002462.
[5] Master in Health and Environment (UFMA), Specialist in Quality Management in Health (Unyleya), Specialist in Clinical Analysis (Florence Institute), Specialist in Clinical Cytology (Laboro), Graduated in Pharmacy (UFMA). ORCID: https://orcid.org/0000-0003-0899-6845. Currículo Lattes: http://lattes.cnpq.br/9904392880678869.
[6] Doctor. ORCID: https://orcid.org/0000-0003-1698-9418.
[7] Medical Residency, Masters, Doctorate. ORCID: https://orcid.org/0000-0002-5460-8594.
[8] PhD in Biotechnology by the Northeast Biotechnology Network-RENORBIO (2011). Graduated in Veterinary Medicine from UEMA (1985), Specialist in Public Health from UNAERP (1994) and Master in Health and Environment from UFMA (2004). ORCID: https://orcid.org/0000-0001-7881-7457. Currículo Lattes: http://lattes.cnpq.br/2790803522614331.
[9] Doctorate in Pharmaceutical Sciences from UFPE, Master in Pharmaceutical Sciences from UFPE, Graduation in Biological Sciences from URCA. ORCID: https://orcid.org/0000-0002-7655-2974.
Currículo Lattes: http://lattes.cnpq.br/0870091628926903.
[10] Doctor. ORCID: https://orcid.org/0000-0002-2300-5248. Currículo Lattes: http://lattes.cnpq.br/3013333572719007.
[11] PhD in Sciences, Master in Pharmacology, Specialization in Pharmacology, Specialization in Biochemistry, Graduated in Pharmacy. ORCID: http://orcid.org/0000-0003-1539-5888.
Currículo Lattes: http://lattes.cnpq.br/3283574594616838.
[12] Doctorate, Master and Specialization in Pharmacology, Pharmacist, Agricultural Technician. ORCID: http://orcid.org/0000-0002-5498-6499. Currículo Lattes: http://lattes.cnpq.br/4315209704773266.
Sent: March 17, 2023.
Approved: June 22, 2023.
















