ORIGINAL ARTICLE
RODRIGUES, Camila Cristine Torres dos Reis; ARAÚJO, Graziela. Systemic changes associated with cardiopulmonary bypass (CPB). Multidisciplinary Core scientific journal of knowledge. 03 year, Ed. 05, vol. 02, pp. 36-54, may 2018. ISSN:2448-0959, Access link: https://www.nucleodoconhecimento.com.br/health/circulation-extracorporea
ABSTRACT
Introduction: the cardiopulmonary bypass (CPB) made possible new cures in heart disease, but there are complications that can be triggered during or after surgery. Objective: Relate the main systemic disorders associated with cardiopulmonary bypass; identifying the major haematological disorders; defining immunological changes and correlating the renal and pulmonary implications. Materials and methods: this is a review of the scientific literature using databases of SciELO, LILACS and PUBMED covering national and international publications in the period of 1993 to 2014. Results and discussion: despite all the modernity the body recognizes ECC as Tin system, generating an immune response that can evolve quickly to a systemic inflammatory response syndrome (SIRS) and also a hematological response through the blood coagulation, is essential to prevent thrombosis patient anticoagulate during surgery with CPB, the time that the patient during the postoperative period, is susceptible to hemorrhage by several factors. Kidney and lung problems are commonly observed after CPB due a multitude of factors, some cases evolve quickly to Acute Renal Failure (IRA), while the pulmonary dysfunction remains the biggest cause of morbidity postoperatively. Final thoughts: the CEC should be used when it is the only form of operative resource for heart patients, because there are severe complications associated with this technique to increase the rates of morbidity and mortality. However, there is a complex interrelationship of the surgical procedure, the anesthesia and the CEC which makes it difficult to assign all the complications to the exclusive use of the CEC.
Key Words: Extracorporeal Perfusion, Heart-Lung, Systemic Complications.
INTRODUCTION
Heart surgery along with cardiopulmonary bypass since the 20th century was one of the greatest achievements in the area of health, because it brought a breakthrough in medicine, significantly reducing the number of deaths and bringing the possibility for new cures in heart disease 16.
In 1953, John h. Gibbon performed the first successful heart surgery using the technique of cardiopulmonary bypass (CPB). This procedure is intended to take the temporary function of heart and lungs when the patient undergoes a surgical procedure. Thus, a set of machines, devices and circuits keep active blood circulation, while these organs are excluded from circulação43.
According to Lima23 (1997), not all patients undergoing cardiac surgery using extracorporeal circulation technique, because although it is a method that has vastly improved on the results, it is known that certain drawbacks still exist, such as: reaction inflammatory, hemodilution, stroke, morbidity, in addition to increased costs. Therefore, when possible, use other techniques without the aid of the CEC to less aggression to the patient.
With the emergence of the CEC, new studies have appeared linked to a different physiology. However, these skills have benefited not only the cardiovascular surgeries, but also all specialties, obtaining concepts for the care of patients during surgery and including the subsequent post-operatório17 period.
Unfortunately, one of the main features of patients undergoing cardiac surgery with CPB is the ease of the organ systems are affected by changes in the function of the cardiovascular system. A variety of complications can occur after heart surgery, in frequency are found: bleeding, respiratory dysfunction, low cardiac debit, renal dysfunction, and neurological changes infecções43.
Thus, the aim of this study was to relate the main systemic disorders associated with the use of cardiopulmonary bypass; identifying the major haematological disorders; defining immunological changes and correlating the renal and pulmonary implications of patient to cardiopulmonary bypass.
MATERIALS AND METHODS
This is a review of the literature with bibliographic survey using scientific databases of SciELO, LILACS and PUBMED. The items found were read and selected, covering national and international publications in the period of 1993 to 2014.
The keywords used in the search were: extracorporeal perfusion, heart/lung, systemic changes.
THEORETICAL FRAMEWORK
CARDIOPULMONARY BYPASS (CPB)
During CPB, heart pumping functions are performed by a mechanical pump and the functions of the lungs are replaced by a device able to perform gas exchange with the sangue20.
In this context, for understanding of the systemic complications related to this procedure it is necessary the basic understanding on the circuit of the blood during cardiopulmonary bypass.
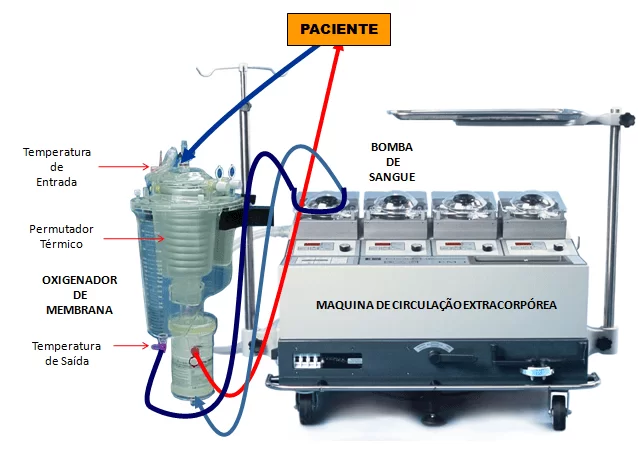
Thus, in CEC, deoxygenated blood is diverted from the heart and lungs to get to the right atrium of the patient, through tubes placed in the superior and inferior Cava vein. By a common thread, the venous blood is taken to the Oxygenator, semi-permeable membrane made reservoir for blood oxygen separation and realization of trade gasosas8.
The Oxygenator, the blood is pumped to a point in the patient’s blood system, usually the ascending aorta, where it travels through the blood system and is distributed to all organs, giving oxygen to the tissues to carry out life processes, and collecting the carbon dioxide in them produced. After circulating through the capillary system of the tissues, the blood back to the system of the upper and lower arches veins, where will be continuously extended to the CEC by the end of the cirurgia14.
For performing cardiac surgery with CPB some procedures have been adopted throughout history to better, as Hypothermia, the use of cardioplégicas solutions and hemodilution.
Hypothermia was associated with extracorporeal circulation with the goal to reduce the metabolic needs of the patients and, therefore, your oxygen consumption, offering additional protection to the body, especially the vital organs, preventing injury from anóxia7. He is currently employed at central hypothermia is induced by cooling the blood with ice water in the heat exchanger of the Oxygenator, so through the cold blood circulation in organs occurs cooling. The reversal of hypothermia, namely reheating the patient gets circulating warm water in calor43 exchanger.
Cardioplégicas solutions, rich in potassium, are used to prevent lesions of the myocardium, leading to near-instantaneous stop of heart, without energy consumption. Alia, for myocardial protection the judicious use of cardioplégicas solutions and hypothermia, so that, after the surgical procedure, occur around the normal state of homeostasis metabólica7.
Before the start of the CEC, to fill the reservoir of the Oxygenator and the other components of the circuit, process known as desareação, use cell phones, not solutions of solutes crystalloids or colloids (called perfusate or prime) thus occurs a dilution of the blood. Studies have shown that this improves oxygenation hemodilution obtained during perfusion, reducing the total amount of blood required to each patient, during and after the operation, preserving a greater number of platelets, as well as reducing blood loss post-operatórias3.
Figure 1: Schematic of the extracorporeal circulation equipment
SYSTEMIC COMPLICATIONS POST CEC
Extracorporeal circulation due to the multiplicity of mechanical components and interactions with the blood produces several changes in the human body that generate large deviations from the physiology, either shortly after perfusion or early post-op 31, 50.
Despite all the modernity of the equipment in use, the longer the time of CEC, the greater the chances of complications. In addition, intrinsic conditions such as age, weight, use of medications, diseases, among others, may compromise the health status of the patient’s end. It is known, for example, that elderly patients and children under the age of three months have worse prognosis, since they are at greater risk for hypothermia, because young children are more prone to heat dispersion and elderly anesthetized the limit for vasoconstriction is the lowest temperature level in adults more jovens24.
Cleveland13 et al. (2001) reported a mortality rate of 2.9% in surgery with CPB and 2.3% no CEC, moreover, operated patients without CEC presented 10.6% of complications, compared with 14% of operated with CEC.
A variety of complications can occur after surgery of the heart and great vessels, some related to the anaesthetic, handling other related to surgery and other related to cardiopulmonary bypass. Often, it is difficult to assign a particular complication to a specific procedure, depending on the interrelation of the procedures in the operating room. Stresses, therefore, that the complications most often found are the immunological complications, hematologic, pulmonary, and renais16.
HAEMATOLOGICAL DISORDERS
The hemorrhagic disorders related to CEC invariably face the changes of blood coagulation, once the blood circulates through pipes and appliances that are non-endothelial surfaces. And, although they are used compatible materials, yet are strange surfaces, able to stimulate simultaneously, to a greater or lesser extent, coagulation and imunológico36 systems.
Occurs, therefore, an imbalance of blood hemostasis during CPB being the most common is the occurrence of thrombotic events, while after the CEC generally are reported bleeding frames.
Thus, during CPB, extrinsic and intrinsic pathways of coagulation cascade will be activated, the first through the factor XII contact to the artificial surface and the second by the release of tissue factor (TF), both culminating in the formation of a clot.
In addition, when the blood is aspirated from the surgical field and returned to give me a by-pass circuit, a series of other elements, such as: fragments of clots and/or fat, tissue thromboplastin, and waste materials, increasing the risk for trombose29.
The CEC induces still changing the fibrinolysis with decrease in formation of plasmin, which favors the emergence of trombos36. In this way, the Association of these factors indicate the importance of patient heparinization right at the start of perfusion to prevent blood clotting by preventing the .36 trombóticos9 events.
The blood thinner heparin is the most widely used for this purpose, because it has few side effects, no inconveniences and there is a specific antidote, protamine, which is used at the end of the perfusion to neutralize the anticoagulant effect of heparin because the post-surgical bleeding can occur because the bleeding effect of heparin. Is valid to mention that simple neutralization heparin stock by appropriate doses of protamine administration restores only partial Hemostatic activity, not being able to recover immediately the hemostatic function of patients undergoing CEC45.
On the other hand, there are several causes for bleeding post CEC, such as: hemodilution, consumption of clotting factors, quantitative and qualitative change of platelets, hypothermia, the host immune response on heparin-protamine complex formation and inadequate surgical hemostasia. Some patients need, therefore, to re-intervention because the bleeding in the early postoperative period and often cannot be identified the cause of bleeding in surgery 1, 28.
The patient during and after CPB may develop thrombocytopenia due several factors, like: deposition of platelets on the internal surfaces of pipes, oxygenators, and filters; result of the action of heparin used for anticoagulation; aggregate capacity reduction due to contact with the surfaces of endothelial no circuits and appliances; dilutional hyponatremia effect on platelets due to hemodilution; In addition to the kidnapping of numerous platelets by the spleen and fígado27.
According to Ascione2 et al. (2001), in prospective studies with random groups of low preoperative risk was found bleeding Framework 1.6 times higher in the group with cardiopulmonary bypass than without cardiopulmonary bypass. It was indicated that less than 20% of patients without CEC needed blood transfusion, compared to more than half of the group with CEC.
Other studies have shown that coronary artery bypass grafting (CABG) without cardiopulmonary bypass has advantages in relation to RM with cardiopulmonary bypass, compared to less postoperative bleeding and less need for transfusion of concentrated of red blood cells, minimizing the morbid factors and costs hospitalares1 .2. So currently, the MRI surgeries are being done without CPB.
It is known that approximately 10 to 20% of patients who undergo the CEC feature excessive bleeding shortly after surgery, about 5 to 10% require in the early hours shortly after replacement surgery sanguínea6.
According to Souza43 et al. (2006) the complex interaction of several factors (hemodilution, hypothermia, trauma, perfusion platelet interaction with endothelial no surfaces, deposition of Fibrinogen in the tubes, reducing the number and activity of circulating platelets and release of a number of platelet activating substances) favor the occurrence of hemostasis and coagulation disorders.
In addition to the own changes mentioned, haemorrhagic CPB also promotes hemolysis of erythrocytes due mainly the deviation from the normal circulation by use of cannulas who form and also by the turmoil centers improper calibration of roller pumps System. However, for surgical teams that use the cardiotomia or cardiotômica suction system, responsible for vacuuming the extravasated blood in the operative field, this practice is the biggest cause of hemolysis, because when the air is suctioned with blood through the vacuum cleaner damage occur to the cells vermelhas11 .51.
Acute hemolysis culminates in hemoglobinemia and hemoglobinuria frames, which can influence the glomerular filtration for obstruction of the glomeruli, glomerular generating a renal34 failure.
IMMUNOLOGICAL CHANGES
When the heart stays without blood during CPB, IE stopped instantly, associated with the interaction with foreign materials to the body occurs as a consequence of the activation of the immune response with possible repercussions for futuras9 complications.
The blood on contact with the artificial surfaces of the perfusion circuit activates the complement system, as well as the inflammatory response. With the complement system activated are released the anafilatoxinas C3a and C5a that stimulate the production of cytokines and several mediators of the inflammatory process. In addition, the white blood cells are also activated by chemotactic power of these anafilatoxinas, with consequent vasocontrição and increased vascular permeability, leading to accumulation of water in the interstício29, 38, 49.
This process is also activated blood clotting and increased neutrophil adhesion to endothelial cells, which is an essential prerequisite to all processes that lead to injury tecidual10.
Thus, neutrophils are activated and release substances that contribute to the formation of generalized inflammatory reaction. During perfusion, neutrophils tend to accumulate in the lungs, where their toxic products produce increased vascular permeability and interstitial edema. In addition, other leukocytes activated release free radicals of oxygen which in certain circumstances can produce microvascular30 occlusion.
This cascade of inflammatory reactions are systemic inflammatory response syndrome (SIRS) and may be manifested in smaller or larger degree, being more evident deleterious and in elderly and neonatos17.
The SIRS also called the pós-perfusão syndrome, clinical features very similar to septic shock. Clinically the SIRS pós-CEC is characterized by impairment of pulmonary function, renal, heart, brain, presence of fever, tachycardia, arterial hypotension, leukocytosis, bleeding disorders, susceptibility to infections, change in vascular permeability leading to accumulation of interstitial fluid, vasoconstricção and hemólise35.
The frequency of clinical manifestations of SRIS after CPB varies between 22% and 27.5%, although it has been assessed by different métodos15. In a 1993 study was reported the incidence of 27.5% of SRIS in children in which the diagnostic criterion used was adapted from definition of SRIS in clínicos41 patients.
In surgeries of RM was observed the SIRS in response body protective around 11%, with a high mortality rate of 41% 25.
Other relevant data, is that the CEC is followed by Acute respiratory distress syndrome (ARDS) with frequency of 0.4% in the adult population, and there is a high mortality associada15, 28.
According to Torrati47 et al. (2012) the CEC produces a systemic inflammatory response in view of the changes in vascular permeability and decrease in oncotic pressure, with release of substances that affect the coagulation and immune response, leading to complications in the immediate postoperative period.
In some patients, the response exacerbated the defense system of the organism occurs due to the release of vasodilator substances such as bradykinin, and your action paralyzes arteriolar vasculature, complicating the output of perfusão29. Because your vasoativo engine occurs increased vascular permeability and arteriolar vasodilation, favoring the extravasation of fluid to the interstice, and bradykinin will remain high during the CEC because your lung site metabolism is excluded from the processo38.
Some patients may develop resistance to the heparin or hypersensitivity reactions caused by heparin-protamine complex, aggravating the protective responses of the imunológico22 system.
A comparative study of MRI with and without CPB identified an inflammatory response by increasing concentrations of C-reactive protein (CRP) and Fibrinogen 12:00 am after the procedure. The RM with CEC biochemical evidence of early pró-trombótico State after surgery, however, no evidence of an increase in the number of thrombotic events. This State can be a consequence of the pró-trombótico give me a by-pass circuit, compensatory response to bleeding or ambos32.
It is worth mentioning that the inflammatory response triggered by the CEC is significantly lower when the infusion time is less than 70 minutos52.
RENAL CHANGES
Kidney failure in the period post CEC is a serious event that is related to the high rate of mortality and morbidity. Despite all the evolution of technology in cardiopulmonary bypass, kidney failure continues showing high incidence, being the development of this complication and multifatorial44 complex.
In the first moments of the CEC’s common framework of hypotension due to a multitude of factors such as: the reduction of perfusion flow hemodilution with reducing blood viscosity, and the increase of the vasodilator bradykinin. After this period, the body begins a compensatory response that often results in hypertension. Thus, the vasoconstriction produced by hypothermia, the elevation of systemic vascular resistance and the absence of circulation pulsatility contribute to this answer hipertensiva43.
However, as a consequence of renal vasoconstriction, occurs a reduction in renal blood flow, predisposing the kidneys to ischemia and injury. In this way, the energy available to the mechanisms of normal kidney activity is reduced, making reabsortivas and secretory functions renais48.
In addition, the hemodilution with crystalloids solutions, when in excess, predisposes the patient to formation of edema, due to reduction of plasma osmotic pressure-coloido, decreasing the reabsorption in capillaries peritubulares, resulting in an aqueous and diuresis rich in electrolytes and can culminate in a hidroeletrolítico39 imbalance.
The hemolysis caused in the CEC produces vasoconstriction by the release of vasoativos products of the interior of the cells and because it is a large molecule, hemoglobin is filtered with difficulty and can crystallize in the kidney tubules, causing obstruction and necrosis tubular34, 37.
Some other factors are also responsible for the production of renal dysfunction, renal disease, including the use of nephrotoxic drugs, renal ischemia agents, severe vasoconstriction, extreme hypothermia and haemodilution profunda44.
In elderly patients the risk of developing acute renal failure disorders more often, because this group of patients most often comes with other diseases such as: diabetes, renal reserve marginal, peripheral vascular disease, and fever rheumatica. So, for a precautionary measure it is important to add mannitol to the perfusate, with the goal of protecting against ischemic injury of the renal47 tissue.
However, there are several evidences that the deleterious effects of cardiopulmonary bypass on the kidneys, including the production of acute renal failure, are related to duration of infusion. This was proven in the study of Taniguchi44 et al., (2007) which identified the negative influence of the time of CEC on renal function by increased serum creatinine, the variation of serum creatinine and a higher incidence of dialysis in patients with CEC time greater than 90 minutes.
The renal insufficiency in heart surgery has an incidence ranging from 3.5% to 31.0%, with higher mortality rate of 0.4% to 4.4% to 1.3% to 22.3%; and the need of dialytic therapy is present in 0.3% to 15.0% of cases, and in these, the mortality rate reaches 25.0% 88.9% 26.
The cardiac surgery with the aid of the CEC feature some typical implications, and some larger and more prevalent than others. Among them is included renal failure with the process of evolution to the acute renal failure (IRA) that occurs on average 7% to 30%. It is believed that genetic factors such as atherosclerosis, diabetes and even hypertension may contribute in renal dysfunction soon after operatório46.
PULMONARY CHANGES
In recent years, due the advancement and the sophistication of equipment and infusion techniques, there was a reduction of pulmonary complications. However, still constitute the most significant cause of postoperative morbidity with such rapid evolution that resists all the measures terapêuticas39.
The patient underwent a heart surgery with CPB usually present a degree of pulmonary dysfunction with decrease of the residual functional capacity of the lungs. Occurs in the lungs, an increase in the interstitial water leakage caused by inflammatory cells, alveolar-filled, leading to inactivation of surfactant and the collapse of some regions, and reduced lung capacity. In addition, exposure to hypothermia during CPB also affects lung function negatively, causing injury to the endothelium pulmonar12.
Another contributing factor to lung dysfunction post CEC are the doses of anesthetics and sedatives that commonly depress ventilation, reducing the residual capacity to work around 20% 5.40.
Thus, the pathophysiology of pulmonary dysfunction is multifactorial, with the combination of the effects generated by anesthesia, surgical incision, ischemia time, surgical trauma on cardiopulmonary bypass, and activation of the immune system. These factors present a great importance on the appearance of respiratory complications in the postoperative period of cardiac surgery 4.21 .33.
Multiple pulmonary complications arise in the postoperative period of cardiac surgery, among which stands out the atelectasis averaging between 60% incidence 90% of the cases, being a number 6 times greater than the heart surgeries without CEC18, 33.
The pulmonary atelectasis is a leading cause of hypoxia that occurs after surgery with CPB, causing a reduction in gas exchange and leading pulmonary problems such as: compression of the lung parenchyma, absorption of alveolar air and commitment surfactante5 .42 function.
Among all the organs affected by the surgery post CEC, the lung tends to be the most committed. Although occurring several factors associated with the increase of interstitial water, the most important is the neutrophil deposition on pulmonary microcirculation, main site of neutrophil sequestration activated. These cells release Lysosomal enzymes and free radicals of oxygen, causing endothelial injury directly and by altering vascular permeability, with consequent accumulation of water in the interstitial pulmonar38.
Studies show the CEC as one of the main causes for the pulmonary injury due to increased airway resistance and possible increase in diaphragmatic dysfunction, and all cardiac surgery raises lung damage, however the CEC is maior21.
However, the study of Montes34 et al. (2004) reports that there was no significant difference in lung function, 72 hours after the surgery, patients operated with and without CPB, both of which had a reduction.
Many studies show that on-call time in the operating room is greater in patients who used the CEC 22.28 .43, 53.. Some authors even claim that patients who use the CEC create greater dependence on mechanical ventilation in post-cirúrgico1 .38. However, this matter is still controversial because there are data that show no significant difference when comparing groups with and without CPB. Similarly, some data indicate that there is no difference in average time to extubation among operated patients with and without CPB 11.29.
FINAL CONSIDERATIONS
Even with the great development of the CEC over its more than 50-year history in Brazil, there are still many implications observed the use of this technique, which despite being the only form of operative resource for many heart procedures can have an impact on severe complications, these being responsible for extending the length of stay with increased hospital costs, as well as important causes of morbidity and mortality.
In large surgeries as there is a complex interrelationship of the cardiac surgical procedure itself, anesthesia and the CEC (especially in relation to the time of surgery), as well as factors related to the patient, and therefore questionable etiology of systemic changes.
REFERENCES
1. ATIK, Fernando Antibas; SAMPATH, Leonardo Augusto; Fábio B. Jatene,; JÚNIOR, José Otávio C. Auler; OLIVEIRA, Sérgio Almeida. The myocardial revascularization without cardiopulmonary bypass minimizes the postoperative bleeding and transfusion need. ARQ. Bras. Cardiol. Vol. 83 No. 4 Sao Paulo Oct. 2004.
2. ASCIONE R, WILLIAMS S, LLOYD CT, SUNDARAMOORTHI T, PITSIS AA, ANGELINI G d. Reduced prostoperative blood loss and transfusion requirement after beating-heart coronary operations: a prospective randomized study. J Thorac Cardiovasc Surg 2001; 121:689-96.
3. ADAMS, James Davis. Immediately after cardiac surgery. Brasília, 2013.
4. ARP AMBROZIN, DI GIACOMO CATANEO AJM. Aspects of pulmonary function after coronary artery bypass grafting relating to preoperative risk. Rev Bras Cir Cardiovasc. 2005; 20 (4): 408-15.
5. AULER JUNIOR, José Otávio Costa; GALAS, Filomena Regina Barbosa Gomes; HAJJAR, Ludhmila Abraham; FRANCA, Zelda. Intraoperative mechanical ventilation. São Paulo, 2007.
6. BARROSO, Roberto Cardoso; MENDONDÇA, Jose Teles de; CARVALHO, Marcos Ramos; Costa Rika Kakuda; SANTOS, José Akil. Evaluation of protamine on neutralization heparin after cardiopulmonary bypass. Rev Bras Cir Cardiovasc vol. 17 No. 1 São Paulo Jan. 2002/Mar..
7. BIAZZOTTO, Camilla B.; BRUDNIEWSKI, Márcio; SCHMIDT, Andrew P.; Junior, José Otávio Auler Coast. Peri-operative period hypothermia. Rev Bras Anestesiol 2006; 56:1: 89-106.
8. BISCEGLI, J. F; ANDRADE, A. J. P; PAULISTA, P. P. DARLINGTON, S. A; SOUZA, L. C. (B); WHEEL, J.; TOFFANO, R. M.; PICCINI, b. clinical evaluation of a membrane oxygenator low filling volume (“PRIMING”). São Paulo, 1999.
9. BRAZIL L; GOMES W J; SOLOMON R; FRANCIS J H P; WHITE J N R, BUFFOLO and corticosteroid use as inhibitor of systemic inflammatory response induced by cardiopulmonary bypass. Rev Bras Cir Cardiovasc 1999; 14 (3): 254-68.
10. BORIWIEC JW, HAGMAN TH, PEKNA M, VENGE P, THELIN s. Circulating cytokines and granulocyte-derived enzymes during complex heart surgery: a clinical study with special reference to heparin coating cardiopulmonary bypass circuits-of. SCAND J Thorac Cardiovasc Surg 1995; 29 (4): 167-74.)
11. CASTRO, Reginaldo Parmar; CROTI, Ulysses Alexander; MACHADO, Maurice of Nassau; MUTILLO, Harold Gonzalez; RINCON Omar Yesid Prieto; Polycarp, Sebastião Rodrigues; FOX, Renata Geron; Braille, Domingo Marcolino. Ultrafiltration technique modification in conventional surgical treatment of congenital heart defects. Rev Bras Cir Cardiovasc vol. 21 No. 1 São José do Rio Preto Jan. 2006/Mar.
12. CLARK SC. Lung injury after cardiopulmonary bypass. Perfusion. 2006; 21 (4): 225-8.
13. CLEVELAND JC, SHROYER LW, CHEN AY, PETERSON AND GROVER, FL. Off-pump coronary artery bypass grafting decreases risk-adjusted mortality and morbidity. Ann Thorac Surg 2001; 72:1282-9
14. COLAFRANCESCHI, Alexander Sicilian; Monteiro, Andrey José de Oliveira; CANALE, Leonardo Secchin; CAMPOS, Luiz Antonio de Almeida; MONTERA, Marcelo Westerlund; SILVA, Paulo Roberto Dutra da; FERNANDES, Marcelo Ramalho; PINTO, Alexander de Araújo; Springs, Stelmar Moura; MOSQUE, Evandro Tinoco. Circulatory assistance with extracorporeal membrane oxygenation (ECMO) in adults: a bankrupt concept or forgotten? ARQ. Bras. Cardiol. Vol. 91 No. 1 São Paulo July 2008.
15. COSTA SOARES, Leonardo Dug; RIBAS, Denise; SPRING, Regine; SILVA, Jean Marcelo Ferreira da; MIYAGUE, Nelson Itiro. Clinical profile of systemic inflammatory response after Pediatric Cardiac Surgery with cardiopulmonary bypass. ARQ Bras Cardiol 2010; 94 (1): 127-133.
16. DIENSTMANN, Caroline; CAREGNATO, Rita Catalina Aquino. Extracorporeal blood circulation in heart surgery: a work camp for the nurse. Rev. SOBECC, São Paulo. Jan/mar. 2013; 18 (1): 35-43.
17. SUNDAY, M Braille. Cardiopulmonary Bypass. Rev Bras Cir Cardiovasc vol. 25 No. 4 Sao Jose do Rio Preto Oct. 2010/Dec..
18. FIGUEIREDO LC, AGUILAR S, ABDALA RCS, ABDALA A, GH CAV. CPAP to 10 cmH2O during cardiopulmonary bypass improves post-operative gas exchange. Rev Bras Cir Cardiovasc. 2008; 23 (2): 209-15.
19. FONSECA, J.W. G; BISCEGLI, J. F; WHEEL, J.; POLANSKY, M. R; AKIKUBO, r. Design and development of a thermometer for circulation. II shows SES, São Paulo 2008.
20. GARCIA, Walter J.; SABA, John C.; BUFFOLO, Enio. 50 years of cardiopulmonary bypass in Brazil: Hugo j. Felipozzi, the pioneer of cardiopulmonary bypass in Brazil. Rev Bras Cir Cardiovasc vol. 20 No. 4 Sao Jose do Rio Preto Oct. 2005/Dec..
21. GUIZILINI S, GARCIA WJ, FARESIN SM, BOLZAN DW, ALVES FA, CATANI R, et al. Evaluation of pulmonary function in patients undergoing myocardial revascularization with and without extracorporeal circulation. Rev Bras Cir Cardiovasc. 2005; 20 (3): 310-6.
22. JOÃO Paulo Ramos, David; JAIN, Fernando Would Do. Immediate care in the postoperative period of cardiac surgery. Rio de Janeiro (RJ) 2003.
23. LIMA, Leonardo Edwards. Cardiac Surgery Less Invasive. A phenomenon of fashion or a method of the future? Editorial Comment. Hospital La Pitiè-Paris, France, 1997.
24. WOLF SON, José Glaucus; PIGLET, Maria Claudia de Azevedo; WOLF BOY, Heraldo Guedis; Smith, John Paul Holland; MAGALHÃES, George Aguilar; LEO SON, Carmelo Sen Ram; FEITOSA, José Francisco Acacius; Oliveira Martins; LAVOR, Arnobius; SOARES FILHO, Odair; Borges, Elita; ABREU, José Sebastião de; DIOGENES, Tereza Cristina Pinheiro; BARRETO, José Erirtônio Feat; PAES Junior, José Nogueira. Left coronary revascularization surgery without CEC and no handling of aorta in patients over 75 years. Rev Bras Cir Cardiovasc 2002; 17 (3): 208-214.
25. Luciano Brandão Machado; NEGRI, Elnara Marcia; BONAFÉ, Wanderley Wesley; SANTOS, Luciana Moraes; MALBOUISSON, Luis Marcelo Sá; CARMONA, Maria José Carvalho. Assessment of the levels of Cytokines and Pulmonary function of patients undergoing cardiac surgery with cardiopulmonary bypass. Rev Bras Anestesiol SCIENTIFIC ARTICLE 2011; 61:3: 275-285.
26. BUCKS GJ, BROWN MA, CHAN WY, HORTON D, TREW P, WHITWORTH JA. Acute renal failure following cardiac surgery: incidence, outcomes and risk factors. Aust N Z JMed 1995; 25:284-9.
27. SAMPATH, Leonardo Augusto; ATIK, Fernando Antibas; Moreira, Luiz Felipe; HUEB, Alexander Ciappina; JATENE, Fabio Biscegli; AULER JUNIOR, José Otávio; OLIVEIRA, Sérgio Almeida. Risk factors of postoperative bleeding after cardiac surgery in adult patients. Rev Bras Cir Cardiovasc 2004; 19 (3): 280-286.
28. MILOT J, PERRON J, LACASSE Y, LETOURNEAU L, CARTIER PC, MALTAIS f. Incidence and predictors of ARDS after cardiac surgery. 2001; 119 (3): 884-8.
29. Mota, André Lupp; Rodrigues, Alfredo José; ÉRVORA, Paulo Roberto Barbosa. Cardiopulmonary bypass in adults in the 21st century. Science, art or empiricism? Rev Bras Cir Cardiovasc 2008; 23 (1): 78-92.
50 h MOURA, Hemilton Vieira; POMERANTZEFF, Pablo M.; GARCIA, Walter Joseph. Systemic inflammatory response syndrome in extracorporeal circulation: role of interleukins. Rev Bras Cir Cardiovasc 2001; 16 (4): 376-87.
31. NOGUEIRA, Celia R. S. R.; HUEB, Whady; Michou TAKIUTI, E.; GIRARDI, Nadia B. M.; NAKANO, Teryo; FERNANDES, Fábio; Felipe da s., PAULITSCH, Aécio f. t. GOIS; LEE, Neuza H. M.; STOLF, A. Noedir. Quality of life after Surgical myocardial Revascularization with and without extracorporeal circulation. Original Article. São Paulo, SP – Brazil, 2008.
32. PAULITSCH, Felipe da Silva. Hemostatic changes and myocardial revascularization surgery clinics with and without cardiopulmonary bypass: a randomized prospective study. São Paulo, 2009.
33. PADOVANI, Cauê; CAVENAGHI, Odette Mauad. Alveolar recruitment in patients in the immediate postoperative period of cardiac surgery. Rev Bras Cir Cardiovasc vol. 26 No. 1 São José do Rio Preto Jan./Mar. 2011.
34. BRIDGES, José Carlos Vieira Dorsa; SILVA, Guilherme Viotto Rao; BENFATTI, Ricardo Dhanashree; MACHADO, Natalia Pereira; PONTELLI, Renato; BRIDGES, Jagan Rose Garden Cury. Risk factors in the development of acute renal failure after myocardial revascularization surgery with CPB. Rev Bras Cir Cardiovasc 2007; 22 (4): 484-490.
35. SALLES, M.J.; SPROVIERI, S.R.; BEDRIKOW, R; PEREIRA, BC; CARDENUTO, S.L; AZEVEDO, P.R.; SILVA, T. M; GOLIN, v. systemic inflammatory response syndrome/sepsis .75 review and study of terminology and physiopathology. São Paulo, 1999.
36. SANTOS, Ari-Thaddeus of the Lily; SPLETTSTOSSER, João Carlos; WARPECHOWSHI, Paul; GAIDZINSKI, Mariana Mariz Pinto. Antifibrinolytics and cardiac surgery with cardiopulmonary bypass. Rev. Bras. Anestesiol. Vol. 57 No. 5 Campinas Sept. 2007/Oct..
37. SANTOS, Fernando Oliveira; SILVEIRA, Marco Antonio; MAIA, Roberto Barreto; MONTEIRO, Marcelo Dantas Chao; MARTINELLI, Reinaldo. Acute Renal failure after coronary artery bypass graft surgery with cardiopulmonary bypass-Incidence, risk factors and mortality. Salvador, BA. 2003.
38. SAVARIS, Laurel. Imunoinflamatória Response To Cardiopulmonary Bypass: Current State. Rev. Bras. Anestesiol 1998; 48:2: 126-136 review article.
39. SOARES, Gustavo Mattos Teixeira; FERNANDEZ, Diego Costa de Souza; GONÇALVES, Mariana Paula Cunha; ALVES, Thales George das; DAVID, Phlegon Lee; HENRIQUES, Karina Mabel Castro; RIANI, Larissa Rodrigues. Prevalence of Major postoperative complications in cardiac surgery. Rev Bras Cardiol. 2011; 24 (3): 139-146.
40. SANGANI, Dante Fanganiello; IASBECH, José Alberto; OLIVEIRA, Sérgio Almeida. Postoperative period in adult cardiac surgery. São Paulo, 1998.
41. SEGHAYE MC, DUCHATEAU J, RG GRABITZ, FAYMONVILLE ML, MESSMER BJ, BURO-RATHSMANN K, et al. Complement activation during cardiopulmonary bypass in infants and children: relation to postoperative multiple organ system failure. Thorac Cardiovasc Surg. 1993; 106 (6): 978-87
42. SILVA, Naila Luisa Saiki; PIOTTO, Raquel Ferrari; BABU, Marcelo Adriano Ingraci; CROTI, Ulysses Alexander; Braille, Domingo m. inhaling hypertonic saline as adjuvant of brathing physiotherapy for reversion of atelectasis in postoperative Pediatric Cardiac surgery. Rev Bras Cir Cardiovasc v. 21 n. 4 Sao Jose do Rio Preto/dez.. 2006.
43. Souza, Maria Helena; ELIAS, Decio. Fundamentals of cardiopulmonary bypass. Second Edition, Rio de Janeiro, Brazil, 2006.
44. TANIGUCHI, Fábio Pope; SHARMA, Ademar Rosa de; Martins, Antonio Sérgio. Cardiopulmonary bypass time as risk factor for acute renal failure. Braz J Cardiovasc Surg 2007; 22 (2): 201-205.
45. TANAKA KA, THOURANI VH, WILLIAMS WH, DUKE PG, LEVY JH, GUYTON RA, PUSKAS JD-Anticoagulant heparin in patients submitted to CEC and CEC revascularization surgery, 2007.
46. TEIXEIRA SOARES, Gustavo Mattos; FERNANDEZ, Diego Costa de Souza; GONÇALVES, Mariana Paula Cunha; ALVES, Thales George das; DAVID, Phlegon Lee; HENRIQUES, Karina Mabel Castro; RIANI, Larissa Rodrigues. Prevalence of Major postoperative complications in cardiac surgery. Rio de Janeiro, 2011.
47. TORRATI, Fernanda Gaspar; DANTAS, Rosana Ap. Spadoti. Cardiopulmonary bypass and complications in the immediate postoperative period of cardiac surgery. ACTA paul. nurses. Vol. 25 No. 3 Sao Paulo, 2012.
48. UTLEY, J.R. RENAL EFFECTS OF CARDIOPULMONARY BYPASS –. IN GRAVLEE, G.P.; DAVIS, R.F.; UTLEY, J.R. Cardiopulmonary Bypass. Principles and Practice Williams & Wilkins, Baltimore, 1993.
49. Grape, Joaquim Miguel Sennfelt de Sousa. Prospective Randomised Study Of Coronary Surgery With And Without Extracorporeal Circulation. PORTO, 2009.
30 h VEGNI, Ronaldo; BRAGA, Gustavo Ferreira de Fabricio; FREITAS, Marcia; DRUMOND, Luis Eduardo; PENNA, William; Y K, José; NOBLE, Gustavo Freitas; KALICHSZTEIN, Marcelo; JAPIASSÚ, Andre Miguel. Complications after coronary artery bypass graft surgery in elderly patients. Rio de Janeiro (RJ), Brazil, 2008.
51. VIEIRA JUNIOR, Francisco Ubaldo; VIEIRA, Reinaldo Wilson; COSTA, Eduardo Tavares; ANTUNES, Nilson; PETRUCCI JUNIOR, Orlando; Pedro OLIVEIRA, Paulo Martins; SEN SON, Rua da Mota; VILARINHO, Thanan de Sousa; Severino, Elaine Soraya Babu. Reflow visualization in blood cleaner and cardiotomia reservoir used in cardiopulmonary bypass. Rev Bras Cir Cardiovasc vol. 24 No. 4 Sao Jose do Rio Preto Oct. 2009/Dec..
52. WHITTEN CW, HILL GE, IVY R, PE GREILICH, LIPTON JM. Does the duration of cardiopulmonary bypass or aortic cross-clamp, in the absence of blood and/or blood product administration, influence the IL-6 response to cardiac surgery? Anesth Analg. 1998; 86 (1): 28-33
[1] Student life sciences course
[2] Master Teacher of the course of Biomedicine
Sent: May, 2018.
Approved: May, 2018.















