ORIGINAL ARTICLE
ROCHA, Ana Paula Coelho [1], ZÁU, Aline Sales Mendes [2], SANTOS, Luciana Mendes dos [3]
ROCHA, Ana Paula Coelho. ZÁU, Aline Sales Mendes. SANTOS, Luciana Mendes dos. Giant congenital melanocytic nevus with neurotization: case report. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year. 07, Ed. 01, Vol. 06, p. 123-132. January 2022. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/health/giant-congenital
ABSTRACT
Congenital Melanocytic Nevi (CMN) are rare hamartomas that are characterized by clonal proliferations of neural crest-derived melanocytes, nesting in the epidermis/dermis after their migration during embryogenesis. Clinically, they are pigmented lesions, with well-defined limits, of variable size and appearance. Giant Congenital Melanocytic Nevus (GCMN) is one with twenty or more centimeters in diameter estimated for adulthood. The presence of a GCMN is associated with a higher risk of severe comorbidities such as Neurocutaneous Melanocytosis and Melanoma, and it is also necessary to exclude Congenital Malignant Melanoma, the main differential diagnosis. In this scenario, would the presence of proliferative nodules be a worse prognostic factor? This report aims to present a challenging clinical case of GCMN with rapidly growing proliferative nodules in an 18-day-old newborn. A brief literature review and the compilation of clinical and histopathological data of the patient in question, who was treated at a Dermatology outpatient clinic, were carried out. It is concluded that although there are clinical characteristics that point to a worse prognosis or possible malignancy, a thorough histological interpretation and correct clinicopathological correlation are essential for adequate diagnosis and therapeutic management.
Keywords: congenital melanoma, giant congenital nevus, melanocytic nevus, neurotization, proliferative nodules.
INTRODUCTION
Congenital Melanocytic Nevi (CMN) are characterized by benign clonal proliferations of melanocytes, derived from the neural crest, which undergo changes in maturation and differentiation during the migration phase in the period of embryogenesis. Nevus cells form nests or are dispersed in the epidermis and deep dermis, in any area of the integument. About 1 to 2% of the population exhibits these changes at birth (SCALVENZI, 2012).
For the initial differentiation between CMN and Acquired Melanocytic Nevus (AMN), the size, precocity of the lesion and the histological characteristics of both are taken into account. CMN tend to be larger and are present from birth or, in general, until the first 6 months of life (VIANA, 2013). In histopathology, there are controversies about real indicators of differentiation between congenital and acquired nevi, and it is often impossible to distinguish them. However, unlike the AMN, the CMN are not restricted to the dermo-epidermal junction, they present greater cellularity and affect deeper planes (ZAAL, 2004).
According to Simons (2017), in the histopathology of CMN, melanocyte nests and nevus cells are found dispersed at different depths of the dermis, between collagen bundles, around annexes, nerves and blood vessels, and may infiltrate subcutaneous tissue, muscle and fascia. There is often high cellularity, varying degrees of architectural disorganization and atypia, as well as pagetoid spread. Such findings are commonly found in melanoma, which increases the difficulty of differentiating between the two and can lead to misdiagnosis.
Clinically, they present as brownish-brown plaques, strongly pigmented, with well-defined borders, a homogeneous and frequently nipple-like surface, with areas of hypertrichosis (MADRIGAL DÍEZ, 2015). However, larger nevi may be asymmetrical, with irregular borders and a heterogeneous, rough or nodular surface due to excess melanocytes and neurotization. This term refers to the process of maturation of nevus cells, which occurs from the surface to the depth of the tissue, where they become more spindle-shaped. These resemble the Schwann cells that make up neurofibroma, a papulo-nodular tumor of firm consistency, whose embryological origin is also in the neural crest (SINGH, 2015).
According to Kopf (1979) CMN can be classified by size, being considered small when smaller than 1.5 cm, medium if between 1.5 and 19.9 cm and giant when greater than or equal to 20 cm, estimated for adulthood. More recently, Krengel (2012) proposed a new classification including, in addition to size, clinical factors with the intention of making descriptions more standardized and assertive regarding the risk of melanoma. In this context, GCMN that present greater roughness, darker pigmentation, hypertrichosis, subcutaneous nodules and presence of satellite nevi are considered to have a worse prognosis, with a higher risk of malignancy.
In this scenario, would the presence of proliferative nodules be a worse prognostic factor? This report aims to present a rare and challenging clinical case of GCMN with rapidly growing proliferative nodules in an 18-day-old newborn, through the exposure of clinical and histological data of the patient in question and a brief review of the literature in order to clarify the relationship between proliferative nodules/neurotization and prognosis.
CASE REPORT
Female NB, 18 days old, born in Amazonas, full-term, weighing 4,415 kg. Mother reports cesarean section due to a high-risk pregnancy, complicated by Preeclampsia, Gestational Diabetes and Repeated Urinary Tract Infection, with multiple antibiotic regimens.
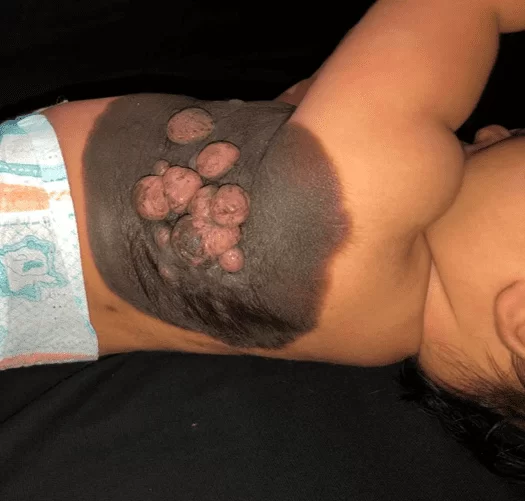
It has a velvety blackened plaque from birth on the left hemithorax, extending from the midclavicular line to the ipsilateral interscapular dorsal region, measuring 23 cm in greatest diameter, with well-defined limits, presence of hair on the periphery, associated with central and coalescent, blackish to pink, ranging from 0.5 cm to 1.5 cm and softened consistency (Figure 1). There is also a satellite lesion measuring approximately 0.7 cm in the left thigh (Figure 2). After 30 days of the initial treatment, there was a significant increase in the mentioned nodules (Figure 3). The rest of the physical examination showed no alterations and the neuropsychomotor development was adequate for the age. There were no suspicious maternal injuries.
After initial dermatological treatment, we opted for an incisional biopsy of a central nodular lesion, due to the clinical aspect suggestive of a worse prognosis. However, the anatomopathological report showed only benign features such as parakeratosis, nests of nevus cells overlying the dermal component of neatly arranged nevus cells, melanic pigment to the deep dermis and areas of neural differentiation, without atypia. Thus, the diagnosis of CMN with neurotization was confirmed and a possible malignant neoplasm was ruled out. Thus, together with the Pediatric Surgery sector, we opted for serial removal of the nodules and clinical follow-up for periodic evaluation of the nevus.
Figure 1. Giant congenital melanocytic nevus: blackened plaque, with a velvety surface, well delimited, hairy, with central tumors, measuring more than 20 cm.
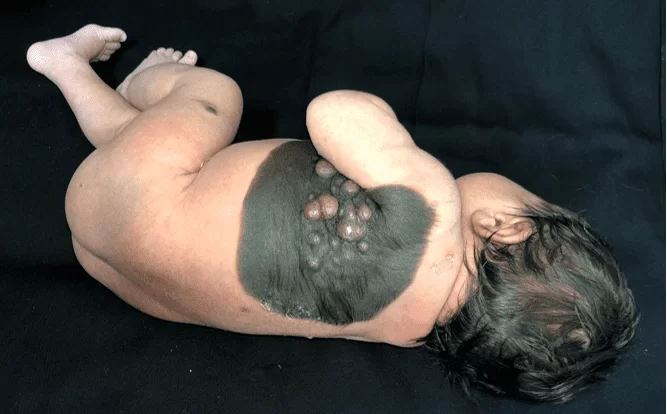
Figure 2. Single satellite lesion in the left thigh.
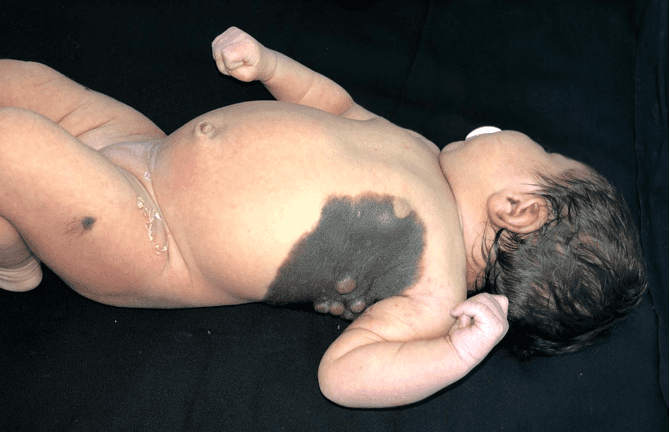
Figure 3. Rapid growth of the lesions 30 days after the initial treatment, forming confluent tumors of softened consistency and pinkish color, maintaining blackened areas on the surface.
DISCUSSION
Giant Congenital Melanocytic Nevus (GCMN) is a rare entity and its average incidence rate is 1 in every 20,000 births (ESCADÓN-PÉREZ, 2019). However, it is of great clinical importance as it is associated with a higher risk of severe comorbidities such as Neurocutaneous Melanocytosis (NMC) and Melanoma (RUGGIERI, 2020). The first is a syndrome that affects the central nervous system and can lead to intracranial hypertension and seizures, especially when the GCMN has nodules, multiple satellite lesions and occurs in locations such as the neck, scalp and dorsal midline. According to Alikhan (2012), the incidence rate of NMC varies between 2.5% and 45%. It is known that melanomas arise primarily from normal melanocytes of the JDE, however, Zaal (2003) points out that up to 11% of GCMN can evolve with malignant degeneration, which occurs in the first decade of life in up to 60% of cases.
The clinical and dermoscopic similarity between GCMN with proliferative nodules and Congenital Malignant Melanoma (CMM), a rare but potentially fatal entity, requires a careful interpretation of the histopathological findings (SCALVENZI, 2012).
This report evidences the presence of a GCMN without significant satellite lesions and with a marked presence of nodules, which is generally present in only 3% of the cases cited in the literature (SCALVENZI, 2012). The anatomopathological findings corroborate the fact that proliferative dermal nodules are not independently indicative of malignancy (SIMONS, 2017). Even so, it finds pairs with regard to the most common location in the trunk, the absence of family and histopathological history favorable to clinical follow-up, seen in 75% of cases (VIANA, 2017).
In this way, there are great questions about the real incidence of Melanoma from cases of GCMN, as well as the need for surgical intervention in the most clinically challenging cases, either because of the size of the lesion or the presence of multiple satellite nevi (VIANA, 2017). There is still no evidence that proves the benefit of this practice as a prophylaxis against the development of malignancy, and, depending on the depth of the nevus cells, there is no possibility of total excision and, therefore, complete cure.
Despite the therapeutic challenge, GCMN still becomes a diagnostic challenge due to its ability to clinically and histologically mimic CMM (ESCADÓN-PÉREZ, 2019).
It is noteworthy that the concomitant presence of multiple satellite nevi should alert to the possibility of meningeal involvement and malignant or benign tumors of the central nervous system. In this scenario, it is necessary to request additional tests, such as magnetic resonance imaging or CSF assessment, for a better evaluation and follow-up of the condition, given its worse prognosis and association with potentially fatal neurological disorders (RUGGIERI, 2020).
Finally, the GCMN is responsible for aesthetic and social damage, generating great emotional impact on those involved (ZAAL, 2012).
FINAL CONSIDERATIONS
In view of the findings in the literature and answering the guiding question, it is concluded that proliferative/neurotized nodules are important in the clinical assessment of the prognosis of GCMN, however, they are not capable of determining a worse outcome in isolation. GCMN present the most varied phenotypes, which vary in terms of risk of developing melanoma or NMC, surgical difficulty and feasibility of exclusive clinical follow-up. Thus, a careful analysis of the set of clinical information combined with the histopathological findings is essential.
These facts illustrate the importance of deepening the study of GCMN, a rare event, however, with the potential to add great morbidity to the patient, as well as to impose obstacles to diagnosis and treatment, which must prioritize the individuality of the case and be prepared in a multidisciplinary way.
REFERENCES
ALIKHAN, Ali; IBRAHIMI, Omar A.; EISEN, Daniel B. Congenital melanocytic nevi: where are we now?: part I. Clinical presentation, epidemiology, pathogenesis, histology, malignant transformation, and neurocutaneous melanosis. Journal of the American Academy of Dermatology, v. 67, n. 4, p. 495. e1-495. e17, 2012.
COTTON, Colleen H.; GOLDBERG, Gerald N. Evolution of congenital melanocytic nevi toward benignity: a case series. Pediatric dermatology, v. 36, n. 2, p. 227-231, 2019.
ESCANDÓN-PÉREZ, Sabrina et al. Nevo melanocítico congénito gigante. Boletín médico del Hospital Infantil de México, v. 76, n. 6, p. 251-258, 2019.
KRENGEL, Sven et al. New recommendations for the categorization of cutaneous features of congenital melanocytic nevi. Journal of the American Academy of Dermatology, v. 68, n. 3, p. 441-451, 2013.
KOPF, Alfred W.; BART, Robert S.; HENNESSEY, Patrick. Congenital nevocytic nevi and malignant melanomas. Journal of the American Academy of Dermatology, v. 1, n. 2, p. 123-130, 1979.
MADRIGAL DÍEZ, C. et al. Nevo melanocítico congénito gigante. Pediatría Atención Primaria, v. 17, n. 68, p. 351-355, 2015.
RUGGIERI, Martino et al. Neurocutaneous melanocytosis (melanosis). Child’s Nervous System, v. 36, n. 10, p. 2571-2596, 2020.
SCALVENZI, Massimiliano et al. Giant congenital melanocytic naevus with proliferative nodules mimicking congenital malignant melanoma: a case report and review of the literature of congenital melanoma. Case reports in dermatological medicine, v. 2013, 2013.
SIMONS, Emily A.; HUANG, Jennifer T.; SCHMIDT, Birgitta. Congenital melanocytic nevi in young children: Histopathologic features and clinical outcomes. Journal of the American Academy of Dermatology, v. 76, n. 5, p. 941-947, 2017.
SINGH, Nidhi et al. Neurotized congenital melanocytic nevus resembling a pigmented neurofibroma. Indian journal of dermatology, v. 60, n. 1, p. 46, 2015.
VIANA, Ana Carolina Leite; GONTIJO, Bernardo; BITTENCOURT, Flávia Vasques. Giant congenital melanocytic nevus. Anais brasileiros de dermatologia, v. 88, p. 863-878, 2013.
VIANA, Ana Carolina Leite et al. A prospective study of patients with large congenital melanocytic nevi and the risk of melanoma. Anais brasileiros de dermatologia, v. 92, p. 200-205, 2017.
ZAAL, L. H. et al. Classification of congenital melanocytic naevi and malignant transformation: a review of the literature. British journal of plastic surgery, v. 57, n. 8, p. 707-719, 2004.
[1] Third-year Dermatology Resident, Bachelor of Medicine. ORCID: 0000-0001-9895-706X.
[2] Third-year Dermatology Resident, Bachelor of Medicine. ORCID: 0000-0002-6561-376X.
[3] Advisor. PhD in Tropical and Infectious Diseases from the State University of Amazonas in partnership with the Tropical Medicine Foundation-FMT-HVD, Specialist in Dermatology, Bachelor of Medicine. ORCID: 0000-0002-3991-6732.
Submitted: January, 2022.
Approved: January, 2022.















