REVIEW ARTICLE
CARNEIRO, Renato Brandi Pereira [1], KFOURI, Flávio de Ávila [2]
CARNEIRO, Renato Brandi Pereira. KFOURI, Flávio de Ávila. D-Ptfe Cytoplast® membranes in guided bone regeneration in implantology. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year 05, Ed. 09, Vol. 05, pp. 108-135. September 2020. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/dentistry/bone-regeneration, DOI: 10.32749/nucleodoconhecimento.com.br/dentistry/bone-regeneration
SUMMARY
The vertical bone increase of alveolar rim is important to obtain good results in rehabilitation with prostheses on implants. This Literature Review sought articles that treat bone increases of alveolar edges using Cytoplast® membranes, seeking to evaluate the resistance to bacterial penetration and capacity to create and maintain space. For this, a literature review was made on the Pubmed and Google scholar search platforms. The researches analyzed found similar results between d-PTFE membrane, titanium meshes and e-PTFE membranes, both in bone gain and bone quality. The space maintenance capacity was evident in the articles in which the titanium reinforced membrane was used. The d-PTFE membrane presented a greater capacity of exposure to the oral environment without compromising the graft material. The authors researched in this study found that titanium-reinforced d-PTFE membranes for bone augamble alveolar rim increases are viable and allow a certain period of exposure to the oral environment without graft contamination. Further studies are needed with the Cytoplast® d-PTFE membrane to explore its characteristics with vertical bone augmentation procedures.
Keywords: Artificial membranes, guided tissue regeneration, bone regeneration.
1. INTRODUCTION
Implantodontics revolutionized dentistry, enabling the restoration of the absence of the dental organ, artificially. Often, the Implantodontist, when planning rehabilitation with implants, is faced with little height or width of the remaining alveolar rim, making it impossible to optimally position the implants according to the retrograde planning performed and to continue the planning can use the guided bone regeneration technique (ROG) to obtain a bone increase in the region that will receive the implants.
The 4 biological principles necessary for success in guided bone regeneration (i.e., PAEE) are: “(i) primary wound closure; (ii) angiogenesis to provide necessary blood supply; (iii) creation and maintenance of space and (iv) wound stability” (WANG; BOYAPATI, 2006, p. 8-11)
Urban et al. (2016) described the flap technique where there is release of periosteum made with periosome and radial incisions and flap advancement with jerks in the coronal direction significantly increasing the surface of the flap, allowing filling with biomaterial and obtaining the primary closure of the wound.
Researchers observed that if a membrane is not exposed and remains eventless for 6 to 9 months, complete bone formation is obtained and that when there is failure, most often it was because of membrane exposure and consequent graft contamination. (CUCCHI et al., 2017)
Urban et al., (2017) emphasized that the membrane must be fixed with tongue and vestibular screws so that it is immobile during the vertical ROG process.
Researchers described the 5 characteristics of a membrane for vertical Guided Bone Regeneration: “(i) Biocompatibility; (ii) Creation and maintenance of space; (iii) Occlusivity and selective permeability; (iv) Tissue integration; (v) Clinical management capability.” (NAUNG; SHEHATA; VAN SICKELS, 2019, p. 2)
Many membranes have been proposed for ROG, both absorbable and non-absorbable and can be collagen, human placenta, human pericardium, human tendon, made of polyglycolic acid, polylactic acid, expanded or dense polytetrafluoroethylene and titanium mesh. (NAUNG; SHEHATA; SICKELS VAN, 2019)
Lee et al. (2020) analyzed the effects of L-PRF as a membrane on horizontal ROG in alveoli raised in dogs, after 4 weeks compared the results of L-PRF with other absorbable and non-absorbable membranes. After another 4 weeks of bone healing, the new bone quantity for each group was measured by microscopy and microcomputed tomography, verifying that the L-PRF plays an effective role as a membrane for the lateral increase of the crest.
Salomão et al. (2010) reported two cases using a polypropylene barrier (Bone Heal® – INP, São Paulo) without the association of any filling material (graft or biomaterial implant), was used in order to keep the clot trapped in order to recover alveolar edge architecture and promote bone neoformation, in order to allow the proper installation of an osteointegrable implant. Before placing the barrier, the alveolo was stimulated to be fully filled with blood.
Non-absorbable membranes, when exposed, have a higher infection rate than absorbable membranes. The dense PTFE membrane prevents the passage of bacteria even when exposed using chlorhexidine mouthwash (0.12%) and applying chlorhexidine gel (1%). (GHENSI et al., 2017)
Patent US7296998B2 of 2007 describes a sheet of hydrophilic polytetrafluoroethylene polytrafluoroethylene (PTFE) polymeric material, which is substantially unexpanded, has cavities that have a depth smaller than the thickness of the sheet. (CHADDICK; BARTEE, 2007).
2. PROPOSITION
The aim of this research is to review the most recent literature on the use of d-PTFE membrane (Cytoplast® Osteogenics Biomedical, Texas) in guided bone regeneration in implantodontia, addressing its resistance to bacterial penetration and its ability to maintain space.
3. LITERATURE REVIEW
Barber et al. (2007) reported two cases of ROG with simultaneous implant installation and d-PTFE membrane covering (Cytoplast® Regentex GBR-200 or TXT-200 Osteogenics Biomedical, Lubbock, TX) without reinforcement. He reported an ROG technique where he left the membrane exposed without exposing the edges for 6 weeks in a case of extraction and immediate implantation of the upper 1st premolar. In the second case report, a trauma accident maintained the Cytoplast membrane® for 4 weeks, easily removing and obtaining a consistent layer of ostoid tissue in the placement of implants in an anterior maxillary region 5 elements without complications and epithelial migration occurred on the ostoid tissue at week 6, increasing the width of keratinized tissue before the injury suffered. Barber et al. (2007) pointed out that “the density of the d-PTFE membrane prevents the colonization of the host flora and prevents contamination of the biomaterial below the membrane”. He also noticed that with this technique the keratinized mucosa heals by second intention, ending its closure with a larger area. Concluded that as the primary closure on the d-PTFE membrane is not necessary, the surgeon can treat large defects, preserve the interdental papilla and preserve the entire width of the keratinized mucosa, without the concern of contamination or bacterial infection and also that the use of Cytoplast® d-PTFE membrane is the ideal treatment option for alveolos of exodontias without primary closure of the wound , due to the simplicity in its placement and removal without compromising the quality of regeneration. (BARBER et al., 2007)
Hoffmann et al. (2008) investigated the clinical regeneration of alveolos of exodontia, using d-PTFE membranes, without the use of graft material, for this purpose, evaluated 276 alveolos of exodontias in 276 individuals (151 men and 125 women; mean age, 50.2 years; age: 24 to 73 years). The percentage of bone gain obtained after ROG using Cytoplast® membrane, without the use of primary wound closure. Before extraction he made a personalized guide that could be fixed to adjacent teeth with five holes where through periodontal probe could obtain measurements of depth of the alveolo after extraction. After extraction, the flaps were elevated and a d-PTFE membrane (Cytoplast® Regentex GBR-200) without titanium reinforcement was placed on the local extraction. The flaps were repositioned and sutured in place. Primary closure was not obtained on the membranes. The membranes were removed after 4 months. The cemento-enamel junctions of adjacent teeth were used as reference points. The measurements were performed using the guide, after extraction and 12 months after surgery in the same areas, the variables described at the bone level were reported individually for all cases reviewed and also in the groups by factors of interest investigated: single alveolo or side by side, mandible, and region, bone gain in vertical defects were expressed in medium and still performed 10 biopsies of hard tissues 12 months after preservation of the alveolo during placement during placement of the implant, obtaining as a result, significant bone gain of the bone nucleus samples, significant regeneration of the volume of the alveolos that could be observed by histological evaluation, indicating that the newly formed tissue at the extraction sites was mainly bone. It found no influence of gender, smoking <=10 cigarettes, age or clinical bone level on the percentage of bone gain. It concluded that the use of Cytoplast® d-PTFE membrane led to the preservation of soft and hard tissue at exodontia sites.
Regarding Randomized Clinical Trials, according to the 2010 CONSORT statement (SCHULZ; ALTMAN et al., 2010):
When properly designed, conducted and reported, they represent the gold standard in health interventions evaluation. To accurately evaluate a study, readers of a published report need complete, clear, and transparent information about its method, technology, and findings. The Consort 2010 State therefore meets the minimum criteria, although this should not prevent authors from including other information if they consider important. (SCHULZ; ALTMAN, et al., 2010, p. 698)
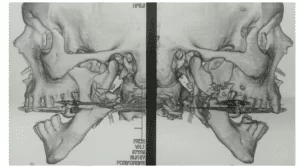
Urban et al. (2014) evaluated the use of the d-PTFE membrane (Cytoplast ® Ti-250) reinforced with titanium in combination with a mixture of autogenous bone and BioOss®, to do this twenty vertical increases in the alveolar ridge using a reinforced d-PTFE membrane with Cytoplast® Ti-250 Osteogenics Biomedical titanium, fixed with Master Pin Control® titanium bone tacks, Messinger and / or Pro-Fix Tentin Screw® tent screws, Osteogenics Biomedical and a 1: 1 mixture of autologous bone and Biooss® and closing without tension with double suture of horizontal and simple mattress, there was no exposure of the membrane. As a result, they obtained a mean bone gain of 5.45 mm and 1.93 standard deviation. Histology revealed that the Biooss® particles “were connected to a dense network of newly formed bone, in all samples, the medullary space was well perfused with blood vessels and no inflammatory response or foreign body reaction was observed.” (URBAN et al., 2017). They concluded that the use of the Cytoplast® Ti-200 membrane is feasible for the technique of increasing the vertical alveolar ridge with autologous bone plus Biooss® 1: 1 and wound closure without tension without exposure of the membrane.
Ronda et al. (2014) carried out a prospective and randomized controlled study designed to test the d-PTFE Cytoplast® Ti-250 and e-PTFE Goretex® membranes, both reinforced with titanium associated with autologous bone and allo-filled in mineralized granules (Puros®, Zimmer Dental, Carlsbad CA, USA), 1: 1 in 23 patients who required vertical bone augmentation of the alveolar ridge with ROG procedures for implant placement in posterior atrophic mandibles (available bone height <7 mm). The implants were placed at the planned level, leaving the apparent turns that were covered by the graft biomaterial, and only then was the membrane used by the surgeon randomized. Wound closure was performed without tension with double suture. The results showed that there was no significant difference between the percentages of bone gain of the two groups. The two biopsies collected in the membrane removal procedure showed two regions: “one of well-organized lamellar bone and another with small gaps hosting osteocytes and a coronal part characterized mainly by bone tissue composed of small and immature trabeculae.” (RONDA et al., 2014, p. 863). They concluded that, the d-PTFE and e-PTFE membranes showed identical clinical results in the treatment of vertical bone defects around the implants, using this technique and that the membrane removal procedure was easier to be performed in the d-PTFE group than that in the e-PTFE group, attributed to the porosities of the outer layer that adhere to the connective tissue, Ronda et al., (2014) emphasized that “although the presence of a porous portion of one of the membranes seems to play an important role in stabilizing the device , favoring its integration with the soft tissue, did not seem to be essential in obtaining bone regeneration. ”
Maridati et al., (2016) proposed and tested the management of d-PTFE Cytoplast® membrane exposures to achieve final clinical success, reported a case, in a procedure of vertical bone augmentation of alveolar rim, a 63-year-old patient where he installed a 3.4Ø x 11 mm implant, leaving some turns and the smooth side of the implant exposed, filled with Biooss® and adapted the membrane reinforced with Cytoplast® titanium fixing with 4 pins , accompanied an exposure of the d-PTFE membrane, even though it closed the wound without tension. There was >3 mm with no sign of infection on the 14th day. The membrane was left in place for another two weeks. In the 4th week the membrane was removed and positioned a palate connective graft to protect the bone graft in the 7th month, the abutman and crown were successfully screwed. Based on the time that due to the Cytoplast membrane having high density and pores smaller than 0,2-0,3 micrometers and having already been tested in alveolos after exodontia before, without primary closure of the wound without tension, in this study obtained good results following the exposure of the membrane > 3 mm and subsequently grafting connective tissue when removing the membrane. They concluded that monitoring the exposure of the membrane without removing it for 4 weeks ensures the maintenance of appropriate space due to the membrane’s resistance to bacterial contamination.
Ghensi et al. (2017) demonstrated that it is possible to handle exposures of the Cytoplast® d-PTFE in their case report, followed < 3 mm exposure of d-PTFE membrane reinforced with autogenous graft and BioOss® 1:1 More tent screw, the primary closure of the wound with release of periosteum without tension was obtained, after 14 days the membrane was exposed, however, there was evidence of an epithelial seal at the site involved, without detaching to probing or suppuration. The problem was solved by continuing mouthwashes with chlorhexidine (0.12%), applying chlorhexidine gel with a concentration of 1% twice a day until the reopening procedure and removing any plaque once a week in the office where it followed for 4 months until removal, resulting in the filling of the Vertical defect of the premolar exodontics region and first upper molar. When the site was reopened, the original defect appeared to have been filled, then two implants (CLC Conic; CLC Scientific, Vicenza, Italy), respectively of size 4 × 10 mm and 5 × 6 mm, were inserted in the positions of the first premolar and molar position. They observed that because non-absorbable d-PTFE membranes present pores 0.2 μm in diameter, prevent bacterial infiltration, and even if the membrane is exposed, the risk of complications and infections is much lower than in e-PTFE membranes. This makes primary soft tissue closure important, but is not strictly necessary, because the membrane functions as an impenetrable barrier to food and bacteria and concluded that d-PTFE membrane exposure can be managed with a deep knowledge of the materials involved and adequate oral hygiene.
Cucchi et al. (2017) evaluated complication rates and vertical bone gain after ROG with Cytoplast® membrane Ti-250 reinforced with titanium compared to titanium mesh covered by collagen absorbable membrane. Comcompared vertical bone gain and complications after ROG in atrophic posterior jaws with Cytoplast® membrane Ti-250XL and titanium mesh of posterior jaws, with placement of implants in 1 stage, analyzed 39 partially toothless patients with atrophic posterior jaw randomly divided into two study groups: 20 patients were treated with one-stage ROG, i.e. with implant placement simultaneously , used cortical perforations to allow angiogenesis and a mixture of autogenous bone and allograft (EnCore®, Osteogenics, Biomedical, Lubbock, Texas), only at this time the surgeon was given the membrane to be used randomly, d-PTFE (Group A) and titanium mesh covered by reticulated collagen membranes (Group B), 19 patients, initially were 40 but 1 patient of this group escaped, were fixed with mini screws and primary closure of the wound without tension with double horizontal and vertical suture. All complications were recorded, distinguishing between “surgical” and “healing” and between “minor” or “major”. Primary implant stability and vertical bone gain were also evaluated. Obtained as results: In group A, the rates of surgical complications, in these cases, temporary paresthesia of the mentual nerve and healing were 5.0% and 15.0%, respectively, In group B, the rates of surgical and healing complications were 15.8% and 21.1%, respectively, were followed for 9 months until removal of the membranes. Bone gain was measured with periodontal probe with divisions of 1 mm and degree of accuracy of measurement of 0.5 mm that were measured from the shoulder of the implant to the bone, before filling with biomaterial by oral, lingual, mesial and distal, in each implant and after regeneration, new measurements were made. And they concluded that the preliminary results of this randomized controlled study showed that d-PTFE membranes and titanium meshes covered by collagen membrane produced similar results both in rate and in types in terms of healing complications. In contrast, d-PTFE membranes showed a lower average rate of surgical complications. Similar vertical bone gain and bone formation were achieved in both groups.
Herzberg (2017) described 10 cases of vertical bone increase of alveolar rim in the region of a tooth, because it is a critical area due to the teeth adjacent to the grafted region, the small flap, being more difficult to adapt to the 3D increase achieved. He made an intrasulcular incision extending up to two adjacent teeth and passing through the crest and mesial relaxant of 10 mm, except when the increase was anterior then the relaxant was performed at the posterior of the incision. He installed an implant in seven patients and three already had implants, and grafted with a 1:1 mixture of autogenous bone with allograft (®, Citagenix) and covered with reinforced d-PTFE membrane (Cytoplast® Ti-250AS, Osteogenics) cutting and adapting, and fixing with self-threading screws (Pro-Fix PFMF®, Osteogenics) and a collagen membrane (Remaix®) was placed on the d-PTFE membrane and closed the wound without tension. After 6 months there was no exposure of the d-PTFE membrane then removed the membrane and of the 10 patients one lost the graft, two lost 0.5 mm and one lost 1 mm, the others had success with no loss. He observed that this technique is reliable and in his references mentions that physiological bone loss of 0.47 to 1.16 mm is common during the first year with a vertical GBR procedure using PFTE membranes can be attributed to the resorption of immature bone in the most coronal part of the regenerated bone preferring implant placement in a second surgery so that this can be corrected at this second moment. It also attributes the success rate to surgical wound closure which in cases of small regions offers smaller flap, more difficult to adapt to the increased environment. It concluded that ROG vertical in region of 1 dental element, using Cytoplast membrane, is a reliable treatment.
Hassan et al. (2017) in its randomized split-mouth study compared the human amnium/cório (MAC) membrane with the Cytoplast membrane® of 22 extraction alveolos of 9 participants in the aspects: amount of preservation of the dimensions of the alveolar ridge, following the ridge preservation procedures, i.e., vertical change of the height of the ridge in millimeters, for this they constructed a radiographic stent with radiopaque reference plane with 5 holes that can be used with millimeter probe and measurement on tomography were made measurements by a professional who was unaware of the objective of the study after the waste and 3.5 months after the waste and the data were used for statistical analysis. It also evaluated bone quality by collecting samples of regenerated bone for histological and histomorphometric analysis and discomfort through the VAS scale. Randomization was performed on the same side or right and left. The results were: Average horizontal increase of 2.54 (1.29) and 2.72 (1.01) for MAC and Cytoplast® respectively, following with a vertical mean increase of: -0.24 (0.91) and 1.18 (2.22) for Cytoplast. It concluded that the mineralized bone area and soft tissue area were not significantly different between the two treatments. Mac sites had significantly lower postoperative VAS scores compared to d-PTFE. The mac exposed intentionally is equally effective in preserving the crest compared to d-PTFE. In addition, the use of MAC can help reduce VAS scores in the postoperative period and potentially result in better bone quality available for implant placement, as evidenced by improved histomorphometric measurements.
Trobos et al. (2018) in their work with in vitro cultures, they evaluated the barrier function of cytoplast®TXT200 d-PTFE and NeoGen® e-PTFE membranes against 2h, 12h and 48h bacteria. His experiment used tubes that had adapted membranes that allowed the contact of a sterile sobreb surface with the inner surface of the membrane and a culture of S. oralis in contact with the external surface and when analyzing the sobrenatant in horse blood agar culture medium. Collecting material from inside the membrane after 48 hours of experiment at 37°C in anaerobiosis condition and there were no bacteria either inside the membranes or in the supernatant in contact with the membranes. However, d-PTFE favored the formation of more Biofilm of S. oralis in relation to titanium and e-PTFE after washing with serum, externally. They concluded that all PTFE membranes were effective barriers against bacterial passage in vitro.
Gallo; Díaz-Báez (2019) with the purpose of describing the management of complications such as exposures and infections after vertical and horizontal ROG with Cytoplast® membrane. Using a new complication management protocol reported for this type of membrane. For this, they evaluated the complications in bone regeneration performed with d-PTFE membrane reinforced with Cytoplast Ti-250 titanium, Osteogenics Biomedical between 2000 and 2017 of 80 cases that had complications and treated cases of membrane exposure less than or equal to 3 mm without infection and after 2 months of surgery keeping for 9 months monitoring the patient’s hygiene weekly by cleaning with chlorhexidine and recommending of slight membrane brushing and application of chlorhexidine in gel to prevent accumulation exposure appeared before that the membrane was maintained for 6 to 8 weeks, following the same hygiene protocol and then removed. When there was a report of pain and pus, chose to immediately remove and cure the granulomatous tissue and keep the tissue hard, the patients were medicated with (amoxicilline/ac. Clavulanic 1g of 12/12h). The sextant with the highest number of complications was the anterior maxilla (43.75%), followed by the left jaw (20%). 70% of cases appeared after 2 months. It concluded that this complication management protocol, which was proposed based on the 80 complications, can help prevent the loss of the entire graft and that infection is the main complication that causes graft loss. It concluded that this proposed new ROG complication management protocol, based on the 80 complications observed, can help prevent total graft loss.
Koidou; Chatzopoulos; Johnson (2019) reported, a series of cases where he used a retiletrated collagen membrane on the alloencherto and on the set fixed a membrane of d-PTFE, which remained for 6 weeks and was atraumatically removed when the d-PTFE membrane was removed, immature connective tissue and it was observed that it became keratinized gum 8 weeks after extraction and concluded that the use of a d-PTFE membrane on a collagen membrane in the post-extraction cavities prevents the degradation of the absorbable membrane and preserves its integrity.
Cucchi et al. (2019) compared rog results between Cytoplastmembrane® and Titanium mesh covered by Osseoguard membrane® did a double blind study comparing the histological and histomorphometric results of bone tissue removed from the grafted region of the hind jaws with the mixture of 1:1 of autogenous bone and allograft (EnCore® Osteogenics), performed decorticalization, using in group A 13 patients with Cytoplast d-PTFE membrane® Ti-250 XL and group B , 12 patients with Titanium Trinon Titanium Titanium mesh®, Karlshure, Germany covered with collagen membrane and fixed with two or three mini profix screws®. Suturou with double horizontal and simple layer, obtained primary closure, waited 9 months. There was no exposure, samples were fixed in formalin phosphate-buferinated followed by hydrochloric acid decalcification and Formic acid, after descaling the samples were dehydrated in series of baths of graduated alcohols and enbedited in paraffin, then the histological sections were corred with hematoxylin and eosin. The slides were scanned (ScanScope CS®, Aperio Tecnologies, Leica Biosystems) in several magnifications to evaluate the characteristics of the new formed bone, the remaining graft material, the integration of grafted material with the new formed bone, soft tissue and local inflammation. Bone regeneration in the coronal part was evident in both groups, and there was intimate contact between grafted material and bone, active bone formation was found was apparent in all regions of grafted bone reflected by high densities of osteocytes and neoformed trabeculated, associated with intense osteoblast activity on bone growth fronts. Osteoclast-mediated resorptions were detected near the graft material, all biopsies contained connective tissue cells with many veins. The grafts were juxtaposed in all samples and there were no inflammatory processes or immunological reactions at the regenerated sites. Histomorphometric data demonstrated both membranes prevented the migration of non-osteogenic cells. Group A n the proportions of bone tissue, graft material and soft tissue were 39.7%; 8,6%; 52.1% and in group B 42.1%; 9.6 and 48.3%, found no significant difference between the groups. It concluded that the results of the study suggested that ROG using Cytoplast® d-PTFE membrane or Ti Trinon Titanium® mesh in combination with autogenous bone and allograft provide similar results both histologically and hitomorphometrically.
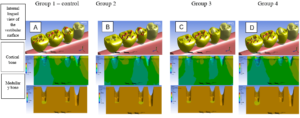
Mertens et al. (2019) tested 4 different in-vitro techniques for volume stability in a wall-edge defect. Group 1: ROG with collagen membrane, particulate xenograft, Group 2: ROG with collagen membrane, particulate xenograft and studs, Group 3: ROG with membrane reinforced with Cytoplast® Titanium Ti-250, particulate xenograft and pins, ROG Group 4 with autogenous block, collagen membrane, particulate xenograft, recorded with tomographies and tested. For this, he created the defects in pig’s jaws and performed the procedures until the flap was closed after he disassembled and made the measurements: before and after the suture, and calculated the change in mm and percentage and found as results that in the 4 groups the flap closure led to a volume reduction, groups 2, 3, 4 had better results than group 1 : collagen without pin. Groups 3 and 4 are better than group 2: collagen and pin and group:3 d-PTFE better result than group 4.They concluded that flap closure has a significant influence on graft stability in 1-wall defects, additional fixation with pins improves ti-d-PTFE membrane stability and block graft showed greater stability than collagen membranes, especially in the shoulder region of the implant, and that defects of a wall graft stabilization are even more important than in auto container defects.
Ibraheem et al. (2020), followed RGB with Cytoplast® membrane with an exposure that developed during the period of graft ossification. To this end, he reported a case of bone augmentation with simultaneous placement of three implants in the region of the teeth 35,36,37, using a d-PTFE Cytoplast® membrane, associated with LifeNet® Health allograft bone, Virginia Beach, VA, around the exposed turns 3-4.5 mm from the buccal side and then completed with a layer of BioOss®, and after that fixed the d-PTFE membrane with ProFix® mini screws. After 21 days the suture was removed and there was an exposure of 3 mm of the membrane, the patient was instructed to pass povidone-iodine twice a day with a cotton wool, the exposure continued to increase, evolving to severe dehiscence in the 16th week, but the edges of the membrane were not exposed, there was no evidence of infection, the membrane was removed revealing fibrous tissue covering the implants. 10 weeks after the membrane was removed, it was reopened and there was regenerated bone covering the cover screws. A core was removed with trephine for histological analysis and hematoxylin / eosin staining slides were prepared, and revealed a viable bone dense and around the bone the integrated allo-fill material. They concluded that the Cytoplast® membrane, when exposed, does not necessarily lead to failure if handled properly with short follow-up intervals.
Mazzucchi et al., (2020) compared samples from the top and bottom of the d-PTFE membrane in relation to bacterial counts of the two surfaces after 28 days of preservation of alveolo of exodontias with exposure to the oral environment. Microbiological and microscopic analyses revealed that a quantity of bacteria was present in the inner layer, below the perialveolar gingival tissues. This finding suggested that bacteria could migrate through the gums adhered to the membrane and colonize the inner part of the membrane, finding a path between the outer layer and gingival tissues that did not adhere to the smooth surface of the d-PTFE membrane. Another possibility is that colonization occurs during surgical procedures: however, in both circumstances, this did not affect the healing process, probably due to the small number of bacteria present at the site.
4. DISCUSSION
Barber et al., (2007) and Ghensi et al., (2017) reported cases of ROG of exodontic alveolos that received biomaterial and the d-PTFE Cytoplast® membrane was used as a barrier. Barber et al. (2007), did not intentionally obtain the primary closure of the wound, which was described by WANG et al. in 2006 as a principle of PASS, claiming to be unnecessary due to the density of the d-PTFE membrane preventing contamination of the biomaterial below the membrane and kept for 6 weeks the membrane exposed to the oral environment with hygiene monitoring and concluded that the primary closure of the wound is unnecessary in his discussion he commented that he and his team did not have enough experience to comment on the effectiveness of keeping the membrane exposed for more than 6 weeks. Ghensi et al., (2017) exactly 10 years later, in his report, despite having double suture and initially obtaining primary wound closure, he reported an exposure of the membrane where he followed for 4 months. Both were successful with ROG, confirming that there was no graft contamination. And Ibraheem et al., (2020) observed a vertical increase exposure for 4 months, and obtained ROG, in this case report the Cytoplast Ti-250 membrane was fixed with screws.
Koidou et al. (2019) in his case series obtained ROG, using Cytoplast® membrane and a collagen membrane between the graft and membrane, leaving the membrane intentionally exposed. Hertzberg (2017) in his case series obtained ROG using the Cytoplast Ti-250 membrane and a collagen membrane over the d-PTFE membrane that was fixed with titanium pins and primary wound closure was obtained.
Hassan et al., (2017) a randomized comparative study model of split mouth was used that decreases bias because it is possible to minimize the interindividual variables, since the participants serve as their own controls and, when present, interfere equally in the treatments. The membranes were Cytoplast® and MAC, ROG was kept maintaining membranes exposed to the oral environment for 3.5 months despite not having obtained as a primary result that the Cytoplast® membrane resisted bacterial penetration mentioned in its conclusion that the MAC exposed intentionally is equally effective in the preservation of the crest compared to d-PTFE and Hoffmann et al., (2008) in its longitudinal retrospective study, ROG performed on 276 alveolos of exodontias, which due to the relatively high population enrolled, reproduces a strong temporal relationship of chance. Some details of the study deserve to be highlighted: first the masking and calibration of the examiner who measured the defects and then the regeneration, and standardized the measurements with individual casquetes allowing 5 measurements mapping each defect, which increases the reliability of the results. When analyzing the data, the variables describing bone loss were reported individually by all cases involved and also separately in arms by investigated factors of interest that were: alveolo format, upper or lower maxillary and region and vertical bone gain was expressed in means ± (DP) and minimum, maximum and mean values. And independent test samples were used to detect some effect related to sex or smoking on bone loss and used Pearson’s correlation to investigate age influence on bone gain. It found no influence of the variables mentioned above. He measured three-dimensional bone gain, obtaining an average vertical bone gain of 6 mm, and also collected samples that demonstrated that there was bone regeneration in histological analysis. In the description of the technique he emphasized that the primary closure of the wound was not obtained, the removal of the membrane was done after 4 months, both authors kept the exposed membranes including MAC for a period between 3.5 and 4 months.
Two experiments have raised relevant evidence on the characteristics of impermeability and integration capacity to Cytoplast® membrane tissues. Trobos et al., (2018) when testing the d-PTFE membrane in its in-vitro experiment with S.oralis culture 2,12,24 and 48hs, concluded that it is impervious to bacteria already Mazzucchi, et al., (2020) analyzed membranes taken from living patients and found bacteria at the bottom of the membrane in small amounts but did not harm the grafts, she argued that they could have penetrated the edges due to poor tissue integration or during surgery.
Gallo et al. (2019) when analyzing the complications in his study of 80 cases, he reported cytoplast membrane exposures®, which followed for 9 months guiding and accompanying patients regarding hygiene and the use of chlorhexidine 0.2% until cure. Proposing a protocol for the management of d-PTFE membrane exposures to the oral environment, it confirmed the resistance to bacterial penetration, corroborating the results and recommendations of Ibraheem et al., (2020).
Ibraheem (2020) in his case report made a ROG in a region of 3 elements, and followed an exposure >3 mm for 10 months and concluded that the Cytoplast® membrane, when exposed, does not necessarily lead to failure if handled appropriately with short follow-up intervals, which confirms that the Cytoplast® d-PTFE membrane resists bacterial penetration corroborating Maridati, et al. , (2016) who had membrane exposure and followed for 4 months and when removing the membrane, also obtained ROG, the latter author did not mention the use of antiseptic but the use of a topical antiseptic seems to play an important role in the prognosis of exposures for Gallo (2019) and Ibraheem (2020).
Urban et al., (2014), in his research, by fixing the reinforced membrane of Ti-d-PTFE Cytoplast® Ti 200 with Studs Master Pin Control®, Messinger and/or tent screws Pro-Fix Tentin Screw®, Osteogenics Biomedical, created the necessary scaffold to create space. When performing cortical perforations, the release of the flap and by obtaining flap closure without tension with double suture of horizontal and simple quilt, there was no exposure of the membrane for 9 months, it met the following principles of Wang’s PAEE, (2006): (i) primary wound closure, (ii) angiogenesis and (iii) space creation. Finding a result of bone gain of 5.45 mm with 1.93 standard deviation confirmed that the ability to create and maintain space and stabilization of the wound were met because if the Cytoplast® d-PTFE membrane did not have the ability to create and maintain space and stabilize the wound in vertical bone increases, it would not have reached the bone increases obtained by the authors , considering that angiogenesis and primary closure were achieved when they described that there was no exposure of the membrane already Mertens et al., (2019) observed in their study with pig mandibles that the act of closing the flaps without tension with double suture, caused a decrease in the vertical volume of the particulate graft and also observed that the use of Studs reduced the deformation of the particulate graft and Herzberg et al., (2017) recognized that often the primary closure of the wound is difficult to be obtained as for example in regions of a tooth.
Ronda et al., (2014) obtained similar results with two different membranes Ti-d-PTFE and Ti-e-PTFE in ROG, 116.8% and 119.7%, there was no exposure of membranes, implants were installed simultaneously with ROG in 1 stage and decorticalization was performed. It is possible to notice that all the principles of the PAEE were fulfilled in this study and Cucchi, (2017) in his comparative study between Cytoplast® reinforced Ti-d-PTFE membrane and titanium mesh covered with collagen membrane for ROG of posterior jaws and placement of 1-stage implants, obtained similar results in bone gain. Still Cucchi, (2019) continuing the work of 2017 with histological and histomorphometric analyses showed evident bone regeneration in the coronal parts of both groups and intimate contact between bone graft material and the graft served as a bridge to the neoformed bone, determining that there was bone regeneration both randomized and masked studies both in the distribution of patients and in the measurements.
Oliveira (2015) in his article on Reports and case series noted that despite their importance, reports and series of cases are at risk of bias and that they usually publish success cases. Because they had a low risk of bias, the following randomized trials: Ronda (2013) and Cucchi (2017) were selected to be grouped into a “Forest Plot” chart. Iportante emphasizes that, in so that they could have been published in the journal Clínical Oral Implant Research, as a consultation conducted on the journal’s submission page, the excerpt follows: “Randomized clinical trials should be reported using CONSORT guidelines. A CONSORT 2010 checklist should also be included in the submission material” (AUTHOR GUIDELINES: 15/09/2020), so once they meet randomized testing guidelines, having met the requirements of the checklist, they can be considered gold standard. They were grouped in a “Forest plot” obtained with the use of the Review Manager 5.4 software that could be performed due to the low heterogeneity between the studies and quantitative results of bone gain in mm, which could be observed by the x² and I² test presented in the subtitles with results 0.55 and 0% respectively, which suggested that the fixed effect analysis model should be used. It only confirms the authors’ conclusions, but demonstrates a pattern tending to favor the Cytoplast membrane® in relation to titanium screen and e-PTFE membrane in bone gain / is understood as capacity for creating and maintaining space also due to the graft relying completely on the membrane to remain position, for example if the studies had twice the total of interventions would already move the black rando out of axis 0, which also proposes to carry out studies such as these with a minimum sampling of two times higher. Review Manager 5.4 software was used to set meta-analysis and simulations.
Figure 1. Forest Plot Average bone gain d-PTFE x Control
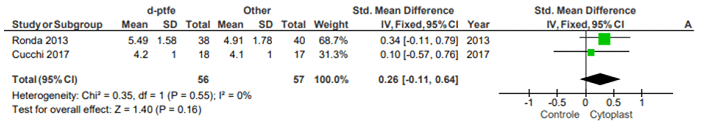
The way Review Manager 5.4 performed the calculations was described by Deeks and Higgins, (2010). The types of data found in the results of randomized clinical trials were continuous, the analysis method chosen was fixed-effect inverse variance and the effect measurement in standard mean difference and 95% confidence intervals.
Analyzing the reports and case series where they chose to try to leave the membrane intentionally exposed, based on the characteristic of impermeability to bacteria and integration of tissues to the membrane and which obtained bone regeneration and bone gain both in height and width and still some proved by histological and histomorphometric analyses, there was still a gap on the quality of the integration of the Cytoplast® d-PTFE membrane to the tissues, if it is complete enough to prevent the flap of the mucosa from allowing the passage of bacteria in cases of exposure, the use of chlorhexidine in topical gel and mechanical removal of plaque in the upper part of the membrane are essential in the control of exposures and periodontal control prior to and after surgical intervention are practices that can minimize this risk. It was observed that there is evidence and that it requires models of studies with this membrane that explore these properties.
5. CONCLUSION
After analyzing the scientific work of this review, we conclude that the Cytoplast® d-PTFE membrane meets the needs of a membrane for preservation and increase of alveolar rim, allowing a period of exposure to the oral cavity, without infiltration of microorganisms in the grafted region provided that the patient is accompanied by a rigid hygiene protocol and the edges of the membrane are covered by tissue and in cases of vertical augmentation when reinforced with titanium and fixed by studs screws, Cytoplast Ti-250 is able to create and maintain space so that there is ROG when obtained closure of the primary wound without tension done with periosteum release incision maneuver.
REFERENCES
BARTEE, Chaddick M.; BARTEE, Barry K.. Hydrophilic high density PTFE medical barrier. Depositante: Chaddick M. Bartee, Barry K. Bartee. US7296998B2. Depósito: 21 set 2007.
BARBER, Dexter H.; LIGNELLI, John; SMITH, Brian M.; BARTEE, Barry K. Using a Dense PTFE Membrane Without Primary Closure to Achieve Bone and Tissue Regeneration. Journal of Oral and Maxillofacial Surgery, Estados Unidos, v. 65, n. 4, p. 748–752, abr. 2007. ISSN: 0278-2391. DOI: 10.1016/j.joms.2006.10.042.
CUCCHI, Alessandro; VIGNUDELLI, Elisabeta; NAPOLITANO, Aldo; MARCHETTI, Claudio; CORINALDESI, Giuseppe. Evaluation of complication rates and vertical bone gain after guided bone regeneration with non-resorbable membranes versus titanium meshes and resorbable membranes. A randomized clinical trial. Clinical implant dentistry and related research, Estados Unidos, v. 19, n. 5, p. 821–832, out. 2017. ISSN: 1523-0899. DOI: 10.1111/cid.12520.
CUCCHI, Alessandro; SARTORI, Maria, PARRILLI, Annapaola; ALDINI, Nicolò N.; VIGNUDELLI, Elisabetta; CORINALDESI, Giuseppe. Histological and histomorphometric analysis of bone tissue after guided bone regeneration with non-resorbable membranes vs resorbable membranes and titanium mesh.(Report). Clinical Implant Dentistry and Related Research, Estados Unidos, v. 21, n. 4, p. 693, ago. 2019. ISSN: 1523-0899. DOI: 10.1111/cid.12814.
DEEKS, Johnathan J.; HIGGINS, Julian P. T. Statistical algorithms in Review Manager 5: on behalf of the Statistical Methods Group of The Cochrane Collaboration, Cochrane Collaboration. p. 1–11, ago. 2010. Disponível em https://training.cochrane.org/handbook/statistical-methods-revman5. Acesso em: 08/07/2020.
GALLO, Pier; DÍAZ-BÁEZ, David. Management of 80 Complications in Vertical and Horizontal Ridge Augmentation with Nonresorbable Membrane (d-PTFE): A Cross-Sectional Study. The International journal of oral & maxillofacial implants, Estados Unidos, v. 34, n. 4, p. 927–935, jul. 2019. ISSN: 0882-2786. DOI: 10.11607/jomi.7214.
GHENSI, P;STABLUM, W; BETTIO, E; SOLDINI, M. C.;TRIPI,T. R.;SOLDINI, C. Menagemente of the exposure of a dense PTFE (d-PTFE) membrane in guided bone regeneration (GBR): A case report. Oral & Implantology, Itália, v. 10, n. 3, p. 335, jul. 2017. ISSN: 1974-5648. DOI: 10.11138/orl/2017.10.3.335.
HASSAN, Muyeenul M.; PRAKASAM, Sivaraman; BAIN, Carol; GHONEIMA, Ahmed; LIU Sean Shih-Yao. A Randomized Split-Mouth Clinical Trial on Effectiveness of Amnion-Chorion Membranes in Alveolar Ridge Preservation: A Clinical, Radiologic, and Morphometric Study. The International Journal of Oral & Maxillofacial Implants, Estados Unidos, v. 32, n. 6, p. 1389–1398, nov. 2017. ISSN: 0882-2786. DOI: 10.11607/jomi.5875.
HERZBERG, RAN. Vertical Guided Bone Regeneration for a Single Missing Tooth Span with Titanium-Reinforced d-PTFE Membranes: Clinical Considerations and Observations of 10 Consecutive Cases with up to 36 Months Follow-up. The International journal of periodontics & restorative dentistry, Estados Unidos, v. 37, n. 6, p. 893–899, nov. 2017. ISSN: 0198-7569. DOI: 10.11607/prd.3380.
HOFFMANN, Oliver.; BARTEE, Barry K.; BEAUMONT, Christian; KASAJ, Adrian;DELI, Giorgio; ZAFIROPOULOS, Gregor Georg. Alveolar bone preservation in extraction sockets using non-resorbable dPTFE membranes: a retrospective non-randomized study. Journal of periodontology, Estados Unidos, v. 79, n. 8, p. 1355–69, ago. 2008. ISSN: 0022-3492. DOI: 10.1902/jop.2008.070502.
IBRAHEEM, Ahmed Gamil; BLANCHARD, Steven B. Alveolar ridge augmentation around exposed mandibular dental implant with histomorphometric analysis. Clinical advances in periodontics, Estados Unidos, jan. 2020. ISSN: 2163-0097. DOI: 10.1002/cap.10090. Disponível em: https://aap.onlinelibrary.wiley.com/doi/epdf/10.1002/cap.10090. Acesso em: 15/09/2020.
KOIDOU, Vasiliki P.; CHATZOPOULOS, Georgios S.; JOHNSON, Deborah. The “Combo Technique”: A Case Series Introducing the Use of a d-PTFE Membrane in Immediate Postextraction Guided Bone Regeneration. J Oral Implantol., Estados Unidos, v. 45, n. 6 p. 486-493, dez. 2019.ISSN: 0160-6972. DOI: 10.1563/aaid-joi-D-19-00015.
LEE, Jun-Beom; LEE, Jung-Tae; HWANG, Sukhiun; CHOI, Jung-Yoo; YEO, In-Sung L. Leukocyte- and platelet-rich fibrin is an effective membrane for lateral ridge augmentation: An in vivo study using a canine model with surgically created defects. Journal of periodontology, Estados Unidos, v. 91, n. 1, p. 120–128, jan. 2020. ISSN: 0022-3492. DOI: 10.1002/JPER.19-0186.
MARIDATI, Paolo. C.;CREMONESI, Sergio; FONTANA, Filippo; CICCIÙ, Marco; MAIORANA, Carlo. Management of d-PTFE Membrane Exposure for Having Final Clinical Success. The Journal of oral implantology, Estados Unidos, v. 42, n. 3, p. 289–291, jun. 2016. ISSN: 0160-6972. DOI: 10.1563/aaid-joi-D-15-00074.
MAZZUCCHI, Giulia; LOLLOBRIGIDA, Marco; LAURITO, Domenica; DI NARDO, Dario; BERLUTTI, Francesca; PASSARIELLO, Claudio; SERAFINI, Giorgio; TESTARELLI, Luca; DE BIASE, Alberto G. Microbiological and FE-SEM Assessment of d-PTFE Membrane Exposed to Oral Environment after Alveolar Socket Preservation Managed with Granular nc-HA. The journal of contemporary dental practice, India, v. 21, n. 4, p. 404–409, 1 abr. 2020. ISSN: 1526-3711.
MERTENS, Christian; SEBASTIAN Braun; KRISAM, Johannes; HOFFMANN, Jürgen. The influence of wound closure on graft stability: An in vitro comparison of different bone grafting techniques for the treatment of one-wall horizontal bone defects. Clinical implant dentistry and related research, Estados Unidos. v. 21, n. 2, p. 284–291, 11 abr. 2019. ISSN: 1523-0899. DOI: 10.1111/cid.12728.
NAUNG, Noel. Ye.; SHEHATA, Ehab.; VAN SICKELS, Joseph. E. Resorbable Versus Nonresorbable Membranes: When and Why? Dental Clinics of North America, Estados Unidos v. 63, n. 3, p. 419–431, jul. 2019. ISSN: 0011-8532. DOI: 10.1016/j.cden.2019.02.008.
OLIVEIRA, Marco Aurélio Pinho; VELARDE, Guillermo Coca ; DE SÁ, Renato Augusto Moreira. Understanding the clinical research V: case report and series of case report. Femina, Brasil, v.43, n 5 p. 235-38, set. 2015. Disponível em: http://files.bvs.br/upload/S/0100-7254/2015/v43n5/a5320.pdf. Acesso em: 05/05/2020. ISSN: 0100-7203
REVIEW MANAGER 5.4 (Rev Man 5). The Cochrane Collaboration, set. 2015. Diponível em: https://training.cochrane.org/online-learning/core-software-cochrane-reviews/revman/reasons-downloading-revman-5. Acesso em: 05/06/2020.
RONDA, Marco; REBAUDI, Alberto; TORELLI, Lucio; STACCHI, Claudio. Expanded vs. dense polytetrafluoroethylene membranes in vertical ridge augmentation around dental implants: a prospective randomized controlled clinical trial. Clinical oral implants research, Dinamarca, v. 25, n. 7, p. 859–866, jul. 2014. ISSN: 0905-7161. DOI: 10.1111/clr.12157.
SALOMÃO, M.; F.K. ALVAREZ; J.T.T. SIQUEIRA. Guided bone regeneration after tooth extraction using membrane exposed to the oral environment. Case reports. Implantnews, Brasil, v. 7, n 6, p. 753-9. jun. 2010. ISSN: 2358-905.
SCHULZ, Knneth F.; ALTMANN, Douglas G.; MOHER, David; CONSORT. CONSORT 2010 Statement: updated guidelines for reporting parallel group randomised trials. International journal of surgery, Inglaterra v.9, n. 8, p. 672-677, 2011. ISSN: 1743-9191. DOI: 10.1016/j.ijsu.2011.09.004.
TROBOS, Margarita; JUHLIN, Annika; SHAH, Furqan A.; HOFFMANN, Maria; SAHLIN, Herman; DAHLIN, Christer. In vitro evaluation of barrier function against oral bacteria of dense and expanded polytetrafluoroethylene (PTFE) membranes for guided bone regeneration. Clinical Implant Dentistry and Related Research, Estados Unidos, v. 20, n. 5, p. 738–748, out. 2018. ISSN: 1523-0899. DOI: 10.1111/cid.12629.
URBAN, Istvan A.; MONJE, Alberto; NEVINS, Myron; NEVINS, Marc L; LOZADA Jamie L; WANG, Hom-Lay. Surgical Management of Significant Maxillary Anterior Vertical Ridge Defects. The International Journal of Periodontics & Restorative Dentistry, Estados Unidos, v. 36, n. 3, p. 329–337, mai. 2016. ISSN: 0198-7569. DOI: 10.11607/prd.2644.
URBAN, Istvan A.; MONJE, Alberto; LOZADA Jamie L; WANG, Hom-Lay. Principles for Vertical Ridge Augmentation in the Atrophic Posterior Mandible: A Technical Review. The International Journal of Periodontics & Restorative Dentistry, Estados Unidos, v. 37, n. 5, p. 639–645, set. 2017. ISSN: 0198-7569. DOI: 10.11607/prd.3200.
WANG, Hom. Lay.; BOYAPATI, Lakshmi. “PASS” principles for predictable bone regeneration. Implant Dentistry, Estados Unidos, v. 15, n. 1, p. 8–17, mar. 2006. ISSN: 1056-6163. DOI: 10.1097/01.id.0000204762.39826.0f.
ONLINELIBRARY.WILEY.COM. Author Guidelines: Research Reporting Guidelines. Clinical Oral Implants Research. Dinamarca. Disponível em: https://onlinelibrary.wiley.com/page/journal/16000501/homepage/ForAuthors.html. Acesso em: 15/09/2020. ISSN: 1600-0501
[1] Graduation in Dentistry.
[2] Advisor. PhD in Science. Professional Master’s degree in Dentistry. Specialization in Implantodontia. Graduation in Dentistry.
Submitted: September, 2020.
Approved: September, 2020.