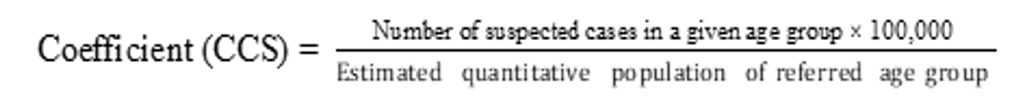ORIGINAL ARTICLE
SILVA, Anderson Walter Costa [1], CUNHA, Arthur Arantes [2], ALVES, Giovana Carvalho [3], CORONA, Rodolfo Antônio [4], DIAS, Claudio Alberto Gellis de Mattos [5], NASSIRI, Reza [6], VEDOVELLI, Silvana [7], VILHENA, Tania Regina Ferreira [8], FAVACHO, Veronica Batista Cambraia [9], SOUSA, Josiany Ferreira [10], ARAÚJO, Maria Helena Mendonça [11], OLIVEIRA, Euzébio [12], DENDASCK, Carla Viana [13], FECURY, Amanda Alves [14]
SILVA, Anderson Walter Costa. Et al. Epidemiologic profile and social determinant of COVID-19 in Macapá, Amapá, Amazon, Brazil. Multidisciplinary Scientific Journal Knowledge Nucleus. Year 05, Ed. 04, Vol. 03, pp. 05-26. April 2020. ISSN:2448-0959, Access link in: https://www.nucleodoconhecimento.com.br/health/covid-19-in-macapa, DOI: 10.32749/nucleodoconhecimento.com.br/health/covid-19-in-macapa
ABSTRACT
In late December 2019, in Wuhan 2019, in Wuhan, China, a cluster of new cases of coronavirus emerged which capable of producing severe Acute Respiratory Syndrome (SARS), The World Health Organization (WHO) declared it worldwide public health emergency. The airway tropism was presumed to be due to the expression of angiotensin-converting enzyme 2 (ACE2). In the North region of Brazil, to date, the state of Amazonas had 26 confirmed cases; Acre 11 cases; Pará 04; Rondônia 03; Roraima and Tocantins 02 cases; and Amapá only 01 confirmed cases. We have quantified and analyzed suspected and confirmed cases of COVID-19 in Macapá, Amapá, Amazonia, which is the first case reports of this region, Brazil. This is an observational, retrospective and quantitative study, referring to the profile of the 108 first suspected cases notified in Macapá, between March 13, 2020, and March 21, 2020, and we attempted to the extrapolate of the incidence of COVID-19 in the 26 Brazilian capitals and Brasília, Federal District, between February 26, 2020, and March 26, 2020. The studies carried out may reveal a pattern of spread of the virus in the community, which warrants the planning and implementation of more effective control strategies and epidemiological surveillance measures.
Keywords: COVID 19, ACE2, coronavirus, pandemic.
INTRODUCTION
In late December of 2019 the end of 2019, in Wuhan, China, new cases of Coronavirus emerged, capable of producing severe Acute Respiratory Syndrome (SARS), which was declared by the World Health Organization (WHO) as a global public health emergency. This virus, called SARS-CoV-2, produces a disease classified as COVID-19 (ICD-10-B34.2) (BRASIL, 2020a; WHO, 2020a; MCINTOSH, 2020). SARS-CoV-2 belongs to the betaCovs genus, of the subfamily Orthocoronavirinae, of the Coronaviridae family. It is an enveloped virus (a double lipid membrane with proteins inserted in it) with a single strand of RNA, which has the shape of a crown, due to the spiked glycoproteins present in its envelope. (CASCELLA, et al., 2020; VELAVAN et al., 2020).
The most common, but nonspecific, symptoms related to infection with this virus are: fever (83.0% -99.0%), dry cough (59.4% -82.0%), dyspnea (55.0%), fatigue (38.1%), sore throat (13.9%) and headache (13.6%) and diarrhea (3.7%) (BRASIL, 2020a; LI et al., 2020; WHO, 2020a). These symptoms can appear, in line with the virus incubation time, on average, 5 to 6 days after infection (WHO, 2020a; ROTHAN et al., 2020).
Airway tropism can be attributed to the expression of angiotensin-converting enzyme 2 (ACE2) in the lung parenchyma, in the epithelium of human airways and in the vascular endothelium. ACE2 is one of the main mediators of virus entry into human host cells, acting as a receptor for pathogen entry. However, this mechanism is not sufficient to explain the affection of human cell lines, which did not express ACE2, but were infected (LI et al., 2020; LI et al., 2003; ROTHAN et al., 2020).
A characteristic of SARS-CoV-2, which contributed to it becoming a worldwide public health problem, is its high rate / potential of transmission, which occurs through fomites (contaminated inanimate materials that serve as vehicles for transmission) and respiratory droplets from coughing and / or sneezing (aerosol transmission). The degree of transmission is dependent upon the level of aerosolized viral load which is estimated to be 3 hours. The oral-fecal route does not seem to be effective, although viable viruses have been found in some cases (CASCELLA, et al., 2020; DOREMALEN et al., 2020; WHO, 2020a). SARS-Cov-2 is third global public health threat following SARS and MERS (NASSIRI, 2020).
According to the Public Health Emergency Operations Center (COE-nCoV) the basic preventive measures are: frequent hand washing using soap and water (for at least 20 seconds); alternatively the use of 70% alcohol or an alcohol-based disinfectant; avoid touching the face, eyes and mouth with unwashed hands; cover mouth and nose with a tissue when sneezing or coughing (and then discard the tissue); clean objects and surfaces touched frequently (door handles, cellphones, remote controls); avoid contact with sick people (isolation); and stay at home if you are showing symptoms, even if you have a common flu (BRASIL, 2020b).
To initiate COVID pneumonia treatment (2019-nCoV), it is dependent on the early recognition of the signs, monitored continuously, considering the clinical manifestations and the general characteristics of the infection. To this end, the Ministry of Health of Brazil created a treatment protocol (BRASIL, 2020b; BRASIL, 2020c).
The high rate transmission factor, combined with the current lack of targeted antiviral therapies of a specific drug for treatment (BRASIL, 2020c), led to the 55,924 cases of COVID-19 confirmed in China, until February 20, 2020 (WHO, 2020b). The fatality, from 44,672 (79.9%) varies according to the age, being 0.2% in patients aged 10-39 years and reaching 8.0% in patients aged 70-79 years and 14.8% in patients aged ≥80 years, the most vulnerable group. The variation in fatality rate can be justified by the associated risk factors, such as previous history of cardiovascular diseases, diabetes, hypertension, chronic respiratory disease and cancer. (GALLASCH et al., 2020; ZHANG, Y. et al, 2020; WHO, 2020a). In Italy, the second most affected country, until March 20, 2020, 47,021 cases of COVID-19 were confirmed, with 4,032 deaths, resulting in an overall fatality of 8.6% (WHO, 2020b).
In Brazil, the first case of COVID-19 was confirmed on February 26, 2020 (WHO, 2020c) and until March 22, 2020, 1,546 cases and 25 deaths were confirmed (22 in the state of São Paulo and 03 in the state of Rio de Janeiro) by COVID-19. Of this total, 926 (59.9%) were registered in the Southeast region; 231 (14.9%) in the Northeast region; 179 (11.6%) in the South region; 161 (10.4%) in the Midwest region; and 49 (3.2%) in the North of the country (BRASIL, 2020d).
In the North region to date, the state of Amazonas had 26 confirmed cases; Acre 11 cases; Pará 04; Rondônia 03; Roraima and Tocantins 02 cases; and Amapá only 01 confirmed cases (BRASIL, 2020d).
Considering the exponential increase of COVID-19 cases and the establishment Brazilian public health challenges, epidemiologic studies that measures the specificities of the population involved and analyzes the influence of social factors on virus transmissibility rate, are of extreme importance and a sanitary necessity (LIPSITCH et al., 2020).
AIM
Quantification and analysis of the first suspected index case and subsequent case conformations of COVID-19 in Macapá, Amapá, Amazonia, Brazil.
Analyze the incidence of confirmed cases of COVID-19 in capitals located in Brazilian Legal Amazon in comparison with the others national capitals and correlate with socioeconomic, sociodemographic and health indicators.
MATERIALS AND METHODS
STUDY PERIODS
This is an observational, retrospective and quantitative study. This study has two time periods. One of them, regarding the profile of the first 108 suspected cases of notification in Macapá, with a limit of March 13, 2020, data from the notification of the first suspected case in Brazil (WHO, 2020c) and March 21, 2020, due to Ordinance nº 454, from the Ministry of Health (MS), which registered the state of community transmission in Brazil (BRASIL, 2020e). It should be noted that cases notified until March 21 may have their verification / release a few days after notification, due or due to laboratory analysis.
The second time phase of this study refers to the extrapolation and calculation of the incidence of COVID-19 in the 26 Brazilian capitals and Brasília, Distrito Federal, is limited between February 26, 2020, date of the first confirmed case in Brazil (WHO, 2020c), and March 26, 2020, one month after confirmation of the first case.
DATA, VARIABLES AND CALCULATION METHODS
This study used two types of secondary data. One of them, referring to the profile of the first 108 suspected cases in Macapá, was the database and aggregated information developed by the Health Secretariat of the Municipality of Macapá, Amapá, Brazil.
The population data, by range, of the municipality of Macapá, were estimated from the Continuous National Household Sample Survey 2016-2018 (BRASIL, 2018a), with reference to the population of Macapá in 2019 (BRASIL, 2020f). These population data were used in the calculation of the Suspected Cases Coefficient (CCS) by age group, regardless of sex, which was calculated according to the formula:
The numbers of confirmed cases of COVID-19, used to calculate the incidences (per 100,000 people) in Brazilian capitals, until March 26, 2020, were extracted from the Coronavirus Map (2020), which provides official and updated aggregated data, provided by the State Health Secretariats, from all Brazilian municipalities with confirmed cases. Population data, used in the calculation of incidences, were extracted from IBGE – Cities and States (BRASIL, 2020f). The incidence calculation method was:
The socioeconomic indicators of the Human Development Index (HDI) and percentage of poor people were extracted from the Atlas Brasil platform (2020). The numbers of health facilities (reference period: October 2015) were extracted from the Ministry of Health (MS) DATASUS platform (BRASIL, 2020g). The demographic density (DD) was calculated using recent information, since the last official is from the 2010 IBGE Census. Thus, the data of the territorial area of the capital (reference year: 2018) and the population of the capital (reference year: 2019) were used to calculate the demographic density (DD = population of the capital in 2019 / area of the capital in square kilometers in 2018).
DATA INCLUSION AND EXCLUSION CRITERIA
For the analysis of the sex of the first 108 records, all suspected cases were included (n = 108); from the time between the first sign / symptom and the notification of the case, eight cases were excluded, five male and three female (n = 100); age, country of residence, number of signs and symptoms, and previous morbidities were excluded in only one case, female (n = 107). As for the trip outside Brazil and contact with a suspected or confirmed case, three cases were excluded, all female (n = 105); as for the reporting health unit, 12 cases were excluded (n = 96); regarding occupation, 3 cases were excluded (n = 105). All exclusions were due to the lack of information (“ignored”). The outliers were not excluded from the analysis.
In the analysis of incidences and correlations, all 26 capitals of the Brazilian Federative Units and Brasília, the federal capital, were included. The capitals were divided into two groups, one of which was composed of Brazilian capitals located in the Brazilian Legal Amazon region (Belém, Boa Vista, Cuiabá, Macapá, Manaus, Palmas, Porto Velho, Rio Branco and São Luís) (BRASIL, 2014) and the other for the remaining 17 capitals plus the federal capital. For the calculation of the incidence, the total number of confirmed cases until 21:00 and 06 minutes of March 26, 2020 was included. Outliers were not excluded from the analysis.
STATISTICAL ANALYSIS
Data were organized, tabulated and analyzed using Microsoft Excel® 2016 software, OriginPro® software version 8.0724 and Statistical Package for the Social Sciences® software version 20.0. It should be noted that the OriginPro® software used did not accept the insertion, in the graphics, of the acute accent.
The distribution of data, when relevant, was tested using the Kolmogorov-Smirnov and Shapiro-Wilk tests. The test was chosen according to the sample size (TORMAN et al., 2012). The homogeneity of variance, when relevant, was tested using the Levene test based on the average (LEVIN et al., 2018).
For the statistical analyzes of the first 108 suspected cases it was used the student’s t test for mean difference of independent samples or U of Mann-Whitney for difference do median. Variables that didn’t have the hypothesis of rejected normal distribution and that showed homogeneity of variance were tested with the t test, whereas variables that didn’t meet these requirements were tested with the Mann-Whitney U test (TORMAN et al., 2012). It was analyzed, comparing the sexes, the significant existence of difference in mean or median of age (in years), the interval of days between the first sign / symptom until the notification date of the suspected case and the quantity of signs / symptoms presented. The level of significance was p-value ≤ 0.05.
The analysis of difference of incidence between the capitals located in the region of the Brazilian Legal Amazon (n=9) and the other Brazilian capitals (n=18) was done without taking into account the hypothesis of normal distribution, due to the sample size being less than 10. Thus, we proceeded to the non-parametric analysis alternative, Mann-Whitney U test. The level of significance was p-value ≤ 0.05. (LEVIN, et al., 2018; TORMAN et al., 2012)
The hypothesis of normal distribution was not rejected for the variables: Incidence of Brazilian capitals, Human Development Index (HDI) of Brazilian capitals, Percentages of poor people in Brazilian capitals and Number of health facilities in Brazilian capitals. Thus, the Pearson correlation test (bivariate) was continued. Correlation directions (positive or negative) and intensity of the Pearson correlation coefficient (r) were observed. The intensity (strong, moderate, weak or nonexistent) is assessed according to the r value (LEVIN et al., 2018). The level of significance adopted was p-value ≤ 0.05.
Descriptive statistics were calculated for: average (?̅), standard deviation (SD), median, minimum and maximum.
LIMITATIONS
Among the limitations of this study are the cases that should not be reported as suspected by the guidelines of the MS (BRASIL, 2020h) but are part of the sample studied. There was also the absence of some information in the database (like occupation, date of notification and first sign/symptom and notifying health establishment), probably due to incorrect or incomplete filling of the notification form.
ETHICAL ASPECTS
The study used aggregate secondary data, that doesn’t allow the individual identification, available in the database of the Municipal Health Secretariat of Macapá, Amapá, Amazonia, Brazil, after institutional consent; in addition to information available at free access electronic addresses. This research followed the criteria of Resolution number 466 of 2012 and number 510 of 2016, of the Brazilian National Council for Ethics in Research (CONEP).
RESULTS
ANALYSIS OF THE FIRST 108 SUSPECTED CASES IN MACAPÁ
Of 108 suspected cases analyzed in this study, confirmed by molecular RT PCR, which was reported between March 13, 2020 and March 21, 2020, three (2.78%) were confirmed with COVID-19. Being all Brazilian, two cases were female (66.66%) and one male (33.33%). The average age of the three cases was 36.3 years. The three (100.0%) had fever, headache, runny nose. Two (66.66%) cases presented with cough, sputum production and difficulty breathing. All three made a trip outside Brazil in the 14 days prior to the notification date. The average time, of the three confirmed cases, between the date of first sign / symptom and the date of notification, was 3 days.
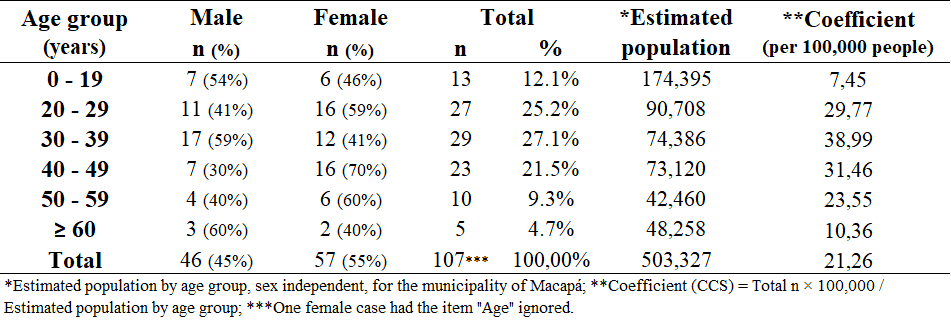
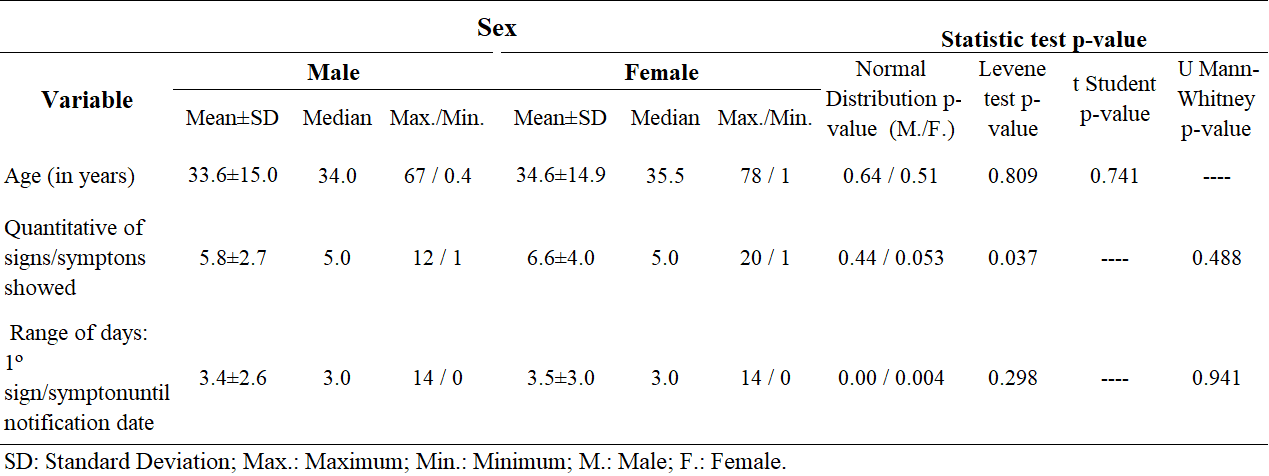
The profile of the total number of Brazilian suspected cases analyzed (n = 107; 99.07%), were as follow: female (n = 59; 54.62%), aged 30-39 years (n = 29; 27, 1%), with no previous morbidities (n = 78; 72.22%) (Table 1 and Table 2 respectively). The average general age (including both sexes), in years, was ?̅ = 34.2 ± 14.9, with a median of 34 years. There was no significant difference in mean between genders (t= 0.331; p = 0.742) (Table 03).
Table 1 Frequency and percentage, by sex and age group, of the suspected cases of COVID-19
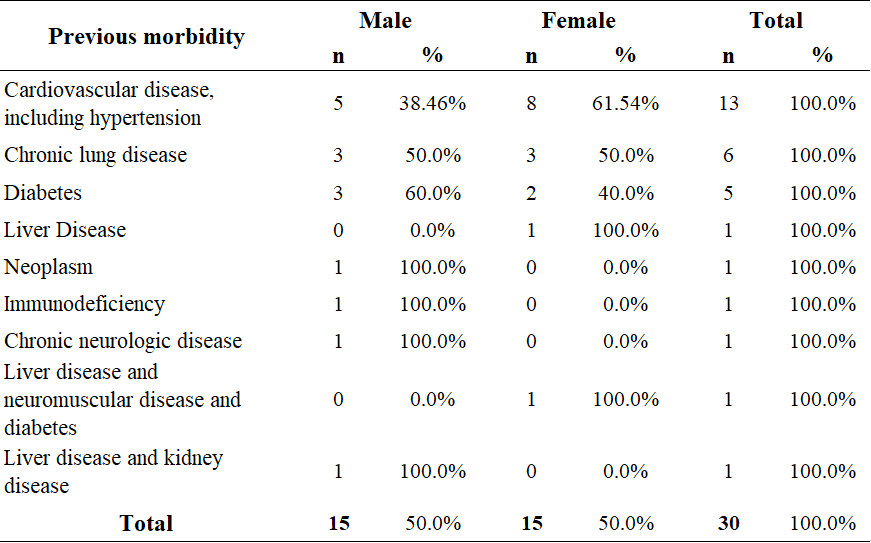
Table 2 Frequency and percentage of previous morbidity of analyzed suspected cases of COVID-19. Macapá, Amapá, 13 to 21 March of 2020.
Table 3 Results of statistic test of analyzed suspected cases of COVID-19. Macapá, Amapá, 13 to 21 March, 2020.
The CCS calculation, through population by age group in the municipality of Macapá, showed that the age group of 30-39 years had the highest CCS, which was 38.99 suspected cases per 100,000 people of that age group. Regardless of age, 21.26 cases per 100,000 people were reported (Table 01).
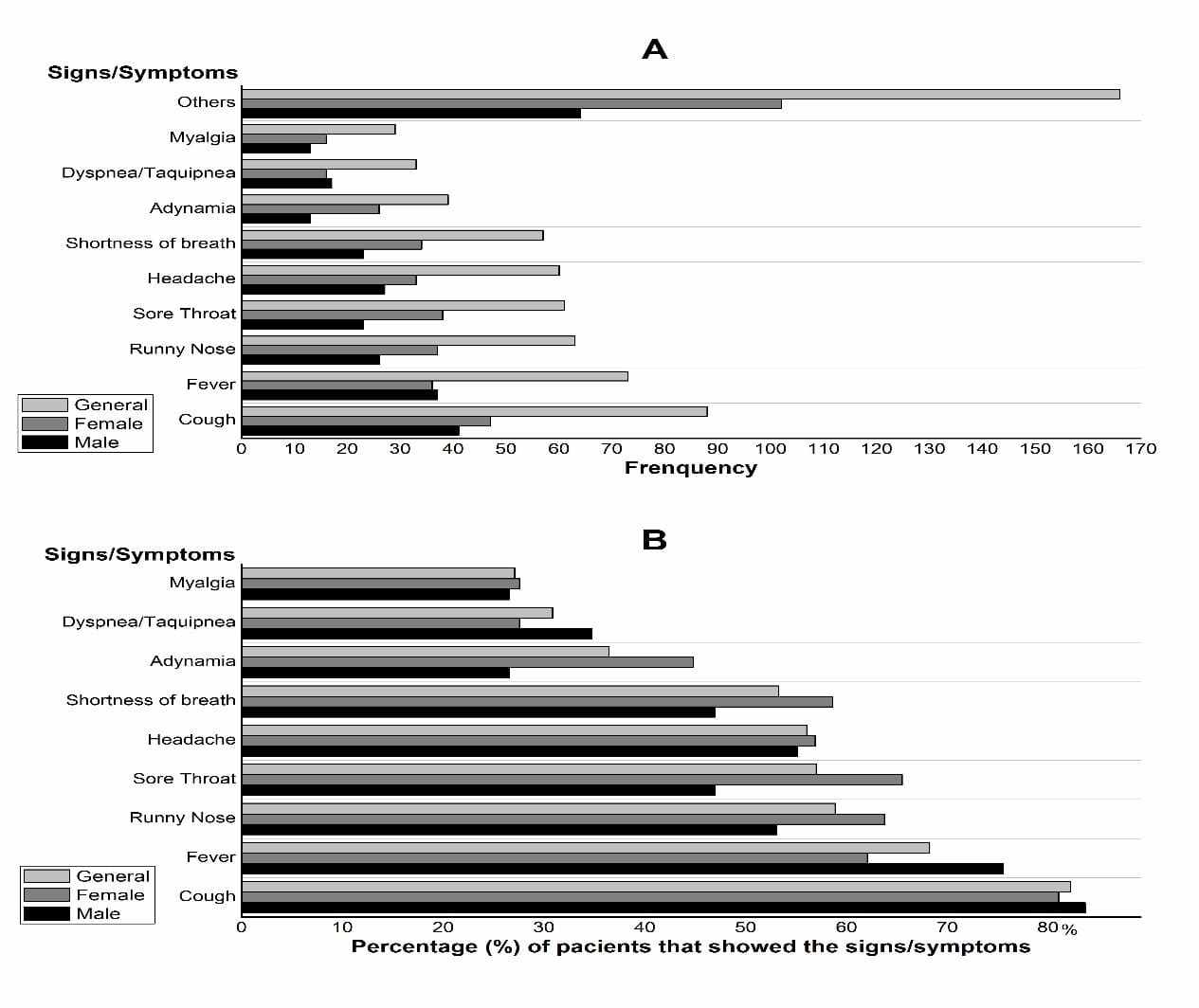
The most frequent signs and symptoms among the suspected cases analyzed, were cough, fever, runny nose, sore throat, headache, shortness of breath, muscle weakness (adynamia), dyspnea, tachypnea and myalgia. Of the pool of this study, 82.2% of patients had cough, 68.2% fever, 58.9% runny nose, sore throat (57.0%), headache (56.1%), difficulty breathing (53.3%), adynamia (36.4%), dyspnea / tachypnea (30.8%) and myalgia (27.1%). From this study, the most common symptom reported was cough. The difference in frequency and percentage between genders can be seen in Graph A and Graph B (Figure 1).
Figure 1 Frequency and percentage of signs and symptoms showed by analyzed suspected cases of COVID-19, by sex and general. Macapá, 13 to 21 March of 2020.
The average number of signs and symptoms per suspected case analyzed, was ?̅ = 6.3 ± 3.5. There was no statistically significant difference between the genders, between the quantitative averages of signs and symptoms presented (U = 1,311; p = 0.488 The time, in days, between the date of the first sign and symptom and the date of notification of the suspected case was ?̅ = 3.4 ± 2.8, with an average of 3 days. There was no significant difference between the gender’s averages in this time interval (U = 1,221.5; p = 0.941) (Table 03).
Only 30 individuals (28.04%), among the suspected cases analyzed, had previous morbidities, of the 107 cases analyzed in this regard. The most frequent morbidity was “cardiovascular disease, including hypertension” (n = 13; 43.33%), followed by “chronic lung disease” (n = 6; 20.00%) and “diabetes” (n = 5; 16.66%) (Table 02).
Healthcare professionals in our study were most frequently exposed (n = 9; 8.57%). However, 88 (83.81%) cases, out of the 105 analyzed in this item, being referred to as “other”. Approximately 81.5% of cases were reported by public health department.
Among the 108 suspected cases, 18 people (16.16%) made a trip outside Brazil, 16 (14.81%) had contact with a suspected or confirmed case of COVID-19 and only 7 (6.5%) were exposed those two conditions. These two conditions were ignored in only three of the 108 cases.
ANALYSIS OF THE INCIDENCE OF CONFIRMED CASES OF COVID-19 IN MACAPÁ AND OTHER CAPITALS
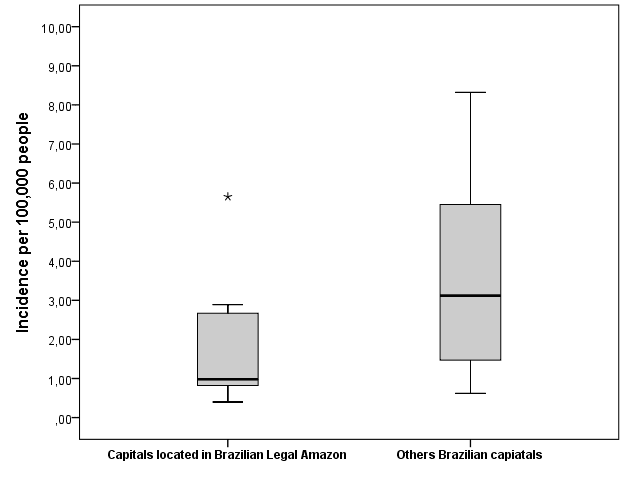
The incidence of confirmed cases of COVID-19 in Macapá, until on March 26, 2020, one month after the first confirmed case in Brazil, was 0.377 cases per 100,000 people. Macapá had the lowest incidence among Brazilian capitals. The average incidence in national capitals was ?̅ = 2.94 ± 2.19 and a median of 2.31. The national capital with the highest incidence was Fortaleza, capital of Ceará, with 8.32 cases per 100,000 people (Figure 02).
Figure 02 Boxplot graph that represents the incidence of COVID-19 in Brazilian capitals, by studied group. Brazilian capitals, Brazil, from February 26, 2020 to March 26, 2020. (*”outlier”, represents the capital Rio Branco, Acre).
The average incidence of the capitals located in the Brazilian Legal Amazon was ?̅ = 1.86 ± 1.70 and a median of 0.98. The capital located in this region, with the highest incidence, was Rio Branco, capital of Acre, with 5.65 cases per 100,000 people (Figure 02). There was a significant difference between the mean incidences of the capitals of the Legal Amazon (n = 9; ?̅ = 1.86 ± 1.70; median=0,98) and the incidences of the other national capitals (n = 18; ?̅ = 3.48 ± 2, 24; median= 3,12) (U = 41.5; p = 0.041).
The incidences in the capitals showed a positive and significant correlation with the demographic density (DD) of the capitals (r = 0.416; p = 0.031), the number of health facilities by capital (r = 0.539; p = 0.004) and with the Human Development Index (HDI) of the capitals (r = 0.436; p = 0.023). A negative and significant correlation emerged with the percentage of poor people by capital (r = -0.409; p = 0.034) (Figure 03).
Figure 03 Graphs of Pearson’s correlation between incidence and: demographic density; the number of health facilities, the Human Development Index and the percentage of poor people. (MCP: Macapá; r: Pearson’s correlation coefficient).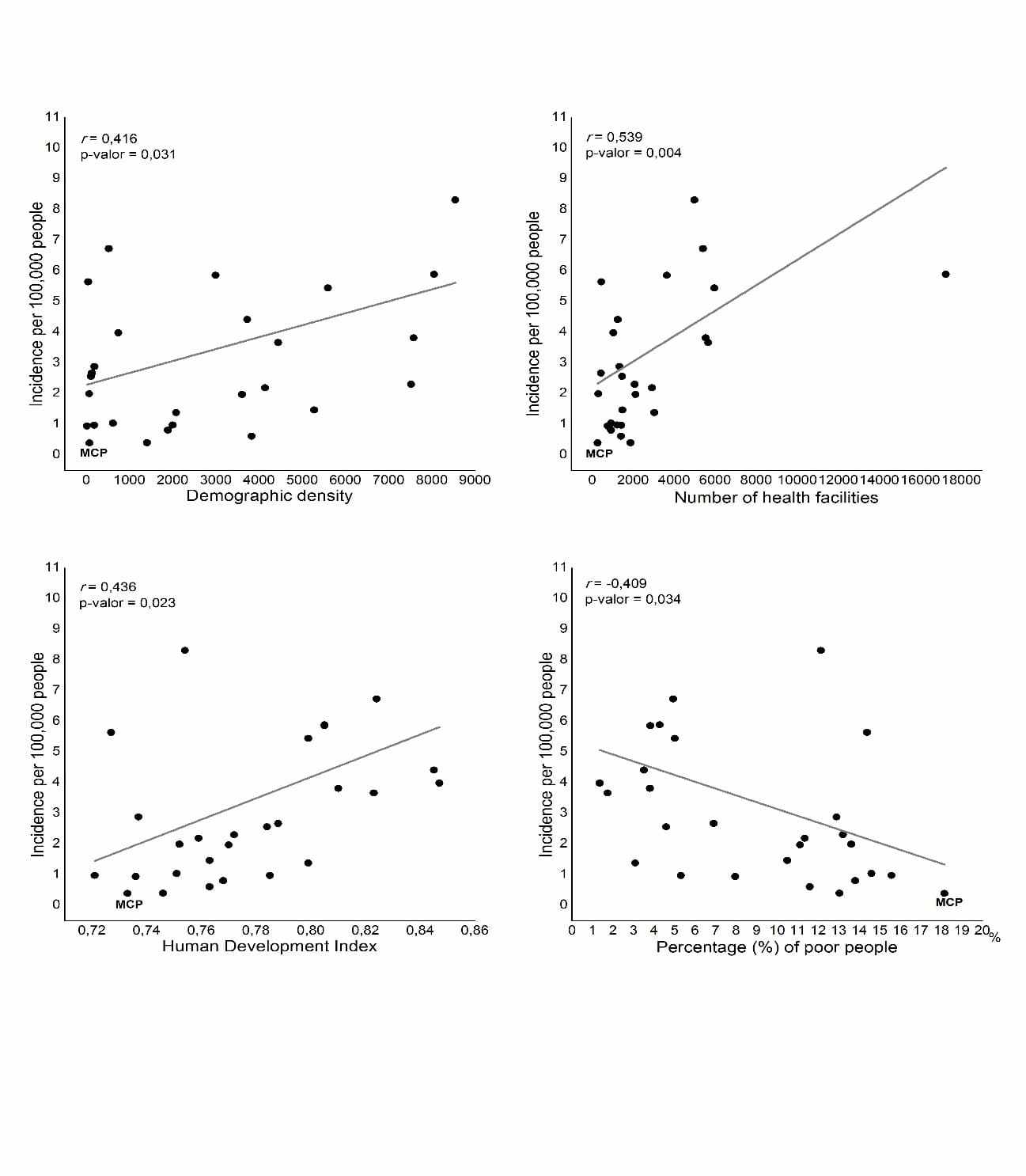
DISCUSSION
The percentage of confirmed cases (n = 3; 2.78%), among the pool of 108 reported as suspected in the municipality of Macapá, was higher when compared to other states, such as Mato Grosso and Minas Gerais, which, until March 20, 2020, had confirmed, respectively, 1 (0.81%) case of 123 notified and 38 (0.92%) cases of 4,122 (SESAMT, 2020; SESAMG, 2020a). The epidemiological profile of the three cases confirmed with COVID-19 analyzed in the present study is similar to the profile of the first four confirmed cases in Minas Gerais, regarding the predominance of the female sex and history of travel abroad in all cases, but there is a difference of approximately 10.5 years on average (SESAMG, 2020b). The average number of days between the first sign and symptom and the notification date of the three confirmed cases was less than one the general average. This reduces the time until diagnosis and treatment. It also reduces the time that the infected patient remains in contact with other people with the possibility of contact and contagion (WHO, 2020a; ZHANG et al., 2020).
The predominance of females (n = 59; 54.62%) in the pool of 108 suspected cases by clinical diagnose of Macapá analyzed in this study, corroborates with the profile of suspected cases reported by SESAMG (2020c) on March 3, 2020 (56.0 % female), with the profile of suspicious cases reported by SESAMA (2020) on March 19, 2020 (62.1% female) and diverges just a little from the profile of the cases in Brazil, reported by the Ministry of Health (MS) on February 10, 2020 (53% male) (BRASIL, 2020b). This predominance can be justified by the slight percentage of women, compared to men, in Macapá (BRASIL, 2018b) and by women being the main public of health services in Brazil (LEVORATO et al., 2014).
The general (male + female) average age of the suspects in the present study, ?̅ = 34.2 ± 14.9 (median = 34 years), is close to the profile in Minas Gerais, 33 years (SESAMG, 2020c). In the State of Ceará, until March 19, 2020, 42.7% of the suspects were female between the ages of 20 and 49 (SESACE, 2020). The similarity between the average age of the sexes was expected, based on the study by ZHANG et al., (2020), which described a ratio of 0.99 male / 1.0 female and a predominance of 89.8% in the age group of 30-79 years of cases in Wuhan, China.
The age groups, regardless of sex, with the highest number of suspected cases reported in the present study was 30-39 years, with 27.1% of cases, very close to the percentage of 20-29 (25.2%). However, even though they had very close percentage, their age groups were distant from each other. Similar and equally balanced percentages for suspected cases were also described in the Epidemiological Report, of March 19, 2020, of the State of Rio Grande do Sul (SESARS, 2020). This similarity between the simple percentage of the age groups of suspected cases can be explained by the percentage distribution of the population, by age group, from Macapá (20-29 = 18.2%; 30-39 = 14.5%) and the State of Rio Grande do Sul (20-29 = 14.0%; 30-39 = 14.6%) (BRASIL, 2018a). And also due to the fact that people in the age range between 26 and 49 years, which include the two age groups 20-29 and 30-39, are the ones who most seek health services in Brazil (LEVORATO et al., 2014).
When analyzing the coefficient of suspected cases of these two age groups, 38.99 (30-39 years) and 29.77 (20-29 years) cases per 100,000 people, calculated in this study, it’s possible to notice that the number of notifications per 100,000 people is 31.0% higher in the 30-39 age group compared to the 20-29 age group. This predominance of the 30-39 years range can be explained because this age group is included in the age range that most seek health services in Brazil (26-49 years), and also the possible relationship between the demand for health services and greater economic laboral productivity. In the State of Amapá, the most productive age group with the largest number of workers (formal employment), 41,365 (31.3%), is 30-39 years (BRASIL, 2018b; LEVORATO et al., 2014). The health demand and access it’s also influenced by income. People with better economic status seek and have better access to health services. In Brazil, the economic status of people with 30 years or older its substantially better when compared to younger age groups (BRASIL, 2018c; TRAVASSOS et al., 2006).
The predominant signs and symptoms investigated in this study were cough (82.2%); fever (68.2%); sore throat (57.0%); dyspnea and tachypnea (30.8%), were described as being the most common nonspecific signs and symptoms in confirmed cases of COVID-19 (BRASIL, 2020a; LI et al., 2020; WHO, 2020a). There was also a concordance with the signs and symptoms of suspected patients described by SESARS (2020) and by BRASIL (2020b). The greatest discrepancy observed was “sore throat”, which presented 57.0% of cases in the current study compared to 13.9% of 55,924 confirmed Chinese cases (WHO, 2020a). This difference may be due to the fact that only 2.78% of the cases analyzed in the present study are confirmed, while all cases described by WHO (2020a) have a confirmed diagnosis of COVID-19. The other possibility is a mixed viral/bacterial infection (ZHANG et al., 2020).
In the current study, the average time, in days, between the date of the first sign and symptom reported by the patient and the date of notification of the suspected case was ?̅ = 3.4 ± 2.8. In 50% of the cases reported as suspected, the delay until notification was greater than or equal to three days (median = 3). This indicates that patients who were considered suspicious for COVID-19 remained symptomatic for an average of 3.4 days, having possible contact with other people. According to WHO (2020a) the transmission of the vast majority of cases occurs by symptomatic patients, although it can rarely occur by an asymptomatic patient, as demonstrated by ROTHE et al. (2020).
The previous morbidities presented by the suspected cases in the present study are similar to those described for 20,812 Chinese patients confirmed with COVID-19, in the study by ZHANG et al. (2020), who described diabetes (5.3%), high blood pressure (12.8%), other cardiovascular diseases (4.2%) and chronic lung diseases (2.4%) as morbidities more associated with COVID-19, with hypertension and other cardiovascular diseases being more related to the unfavorable outcome.
The only professional category mentioned in this study was that of health (n=9; 8.57%), that are part of the main risk groups for COVID-19 (GALLASCH et al., 2020; ZHANG et al., 2020). Moreover, a high number of marks was noted as “other” (n = 88; 83.82%). This can impair the assessment of other possible risk professions, which may be related to a higher risk of infection (KOH, 2020; ZHANG et al., 2020;) The vast majority of suspected cases analyzed in this study were reported by the Unified System (SUS), which can demonstrate the importance and power of this state system, which serves the vast majority of the Brazilian population in a comprehensive and free manner (BRASIL, 2020f; VIANA et al., 2009).
A significant correlation between the incidence of COVID-19 and certain indicators (e.g.: percentage of poor people, quantitative of health units) was identified. At least in the initial stage, until March 26, 2020, of the spread of the virus, there is a correlation between higher values of incidence of COVID-19 and the population residing in cities with good indicators of social development.
In our study, demographic density (DD) emerged as a positive correlate. The highest number of cases of COVID-19 / 100,000 people correlates with a greater number of population density. It’s important to highlight that the agglomeration of individuals makes the spread of the virus easier, since infections in family groups, as well as in health professionals, confirm the occurrence of human-to-human transmission, mostly through close contact (READ et al., 2020; BRASIL, 2020i). In China, the most densely occupied cities, with the highest number of daily flights, were the most affected (LAI et al., 2020; READ et al., 2020). Macapá, the city with the lowest incidence indicated in this study, until February 2020, had communication with daily and direct commercial flights only to Brasília, Distrito Federal, and Belém, Pará (BRASIL, 2020j).
There are still few discussions about population characteristics related to COVID-19. Some studies, such as the one carried out by WHO (2020a), may justify the result of this association, as they show a higher rate of transmissibility in regions with a higher population concentration. In addition, the area of the Brazilian government responsible for health proposes, in a generalized way, that certain outbreaks in areas of high population density have a greater chance of spreading and thus represent events with the potential to cause a great impact on public health (BRASIL, 2009).
In a mapping and spatial analysis study conducted in China, the investigators demonstrated that the distribution of COVID-19 cases was not random. The concentration points of cases at the beginning of epidemic were restricted to areas of greater economic development and population density. In addition to cultural and socioeconomic factors, the disease exposure factors are related to the greater number of cases for offering greater pendular mobility, greater number of trips and greater quantity of health facilities enabled to perform the care and diagnosis (FAN et al., 2020a; LAI et al., 2020; OLIVEIRA et al., 2019; ZASLAVSKY et al., 2017).
Thus, the evidence mentioned above corroborates and justifies the results found in the present study, since certain sociodemographic and health indicators showed a positive and significant correlation with the incidence rate of COVID-19 between the capitals, that is, the quantification and analysis of registered cases, between 26 February and March 26, in Brazil, demonstrated that the capitals that had higher demographic density (DD), Human Development Index (HDI) and more health facilities had higher incidence rates.
Reliable social indicators (e.g., higher DD, HDI and more health facilities) can be attributed to a larger number of fluctuating populations, for example, those that are present in the territory, on a specific date, for a short period and in search of different activities (BRASIL, 2011). Thus, cities with effective social indicators that offer possibilities for tourism and business opportunities may have a higher incidence of infections, especially those transmitted by human to human. Such cities provide more attractions for this seasonal and non-resident population since the individual as this group can serve as potential carrier for the transmission of the virus (FAN et al., 2020b). A correlation study carried out by FAN et al. (2020b) assessed the association between the quantity of the floating population of Wuhan and the number of confirmed cases of COVID-19, which obtained correlation coefficient of 0.84. This indicates that the greater the fluctuating number of people in a region, the higher the number of confirmed cases.
The negative correlation (r=-0.409; p=0.034) between the incidence of COVID-19 and the percentage of poor people by capital, showed that the higher the number of cases/100,000 people, lower is the percentage of poor people living in the referred capital. This result reaffirms the existence of association between incidence and HDI, also observed in this study.
The number of exported infected cases can directly influence the possibility of a larger secondary cases in community and capitals with a higher percentage of disease transmission among poor people who don’t travel, and initially who in principle, show lower risk of exposure (KUCHARSKI et al., 2020; ROSA, 2006). In general, this population has less access to educational, information and health resources. In marginalized population, it would be challenging to recognize the initial COVIND19 signs and symptoms, which can pose errors in case identification in view of the fact they often have limiting access to health facilities. This can lead to underreporting diagnostic cases (CAMPELLO et al., 2018).
In agreement with these observations, the results of the present study demonstrated that Macapá is the capital that has the lowest incidence per 100,000 people of COVID-19 following one month of confirmation of the first case in Brazil, which accordingly, has one of the lowest Human Development Index (HDIs) and one of the lowest demographic density (DDs) among the Brazilian capitals, the lowest number of health facilities in the country, a restricted number of daily commercial flights, and the highest percentage of poor people among all national capitals (ATLAS BRASIL, 2020; BRASIL, 2020g; BRASIL, 2020j).
The incidence until March 26, 2020, in the group of capitals in the Brazilian Legal Amazon region (n = 9; ?̅ = 1.86 ± 1.70; median = 0.98), was lower (U = 41.5; p = 0.041) than the the group of other Brazilian capitals (n = 18 ; ?̅ = 3.48 ± 2.24; median = 3.12). This difference may be due to the socio-spatial isolation of the Legal Amazon, in relation to the rest of Brazil, described by VIANA et al. (2009). An example of this isolation is the capital Macapá, which lives 59.5% of the population of Amapá, which is not connected by land to the rest of the country (BRASIL, 2020f; DRUMMOND, 2000) and shows, in this study, a lower index (0.397) of COVID-19 among Brazilian capitals.
This difference in the incidence of COVID-19 may, in addition to socio-spatial isolation, be related to climatic and urban factors in the Amazon region, such as high relative statistics and demographic density (BRASIL, 2020f; SILVA et al., 2013; WANG et al., 2020).
At first, the large urban centers of the Brazilian Amazon have an history of epidemics of viral diseases transmitted by mosquitoes, such as Zika and Yellow Fever. However, the climatic factors of the Amazon ecosystem that act to support endemic transmission and / or the emergence of epidemic waves, by enabling the reproduction of mosquitoes (FARIA et al., 2018; GIOVANETTI et al., 2020), may favor the transmission of COVID -19 in the Amazon.
In the studies in vitro by SILVA et al. (2013), by WANG et al. (2020) and KAMPF et al., (2020), elevated relative humidity is consistently related to lower viability of enveloped viruses that cause respiratory diseases, such as the coronavirus. This may be due to the lower viral stability at higher temperatures and because the respiratory droplets remain less time suspended in the air with high humidity (WANG et al., 2020). The results by OLIVEIROS et al. (2020) demonstrated that the number of COVID-19 cases, in a linear regression model, suffered only 18.0% of influence due to these climatic factors. While population density, urban transport, cultural aspects, public health policies and remoteness measures influence 82.0%.
This demonstrates that, possibly, only climatic factors will not be enough to contain the increase in the number of cases, requiring other measures, such as social isolation, decreed by the state government in some federative units of the Brazilian Legal Amazon, such as Amapá (AP, 2020; RIPAP, 2020). In the absence of effective vaccines or antivirals, measures such as social withdrawal, have the potential to reduce the eccentricity of the COVID-19 incidence curve, flattening its peak and reducing the acute burden on the health system. It is essential that government authorities pay attention to epidemiological alerts and what the scientific community has to contribute (HAFFAJEE et al., 2020; PREM et al., 2020; RIPAP, 2020; WU et al., 2020).
CONCLUSION
The epidemiological prolife of suspected COVID19 patients presented in this study is consistent with the current literature, although we acknowledge characterization of suspected cases is currently limited. Locations such as Macapá, where presents distinct social, geographical and economic characteristics, could reveal a pattern of COVID-19 transmission, necessitates continuous and rigorous monitoring of the epidemiological profile of cases.
Macapá, when compared to other Brazilian locations, can have a better opportunity to fight the virus. The coordination of case notification can be optimized by effective communication among various stakeholders including health institutions and professional health societies.
The role of government in education of the public about COVID19 awareness is critical in reducing the burden of disease. In addition, registry of the occupational profile of the notified case can be further improved, for example, including moto boys, couriers, and public security servers, who are likely to have more exposure compared to other professionals.
More than 80.0% of analyzed cases in our study were notified by public health units. This demonstrates the importance of a robust health state-owned systems that could make the difference in containing the COVID-19 epidemic, both in Macapá, the Brazilian Amazon and the rest of the country.
The correlation between the incidence of COVID-19 and the social status, sociodemographic and health profile implies that the capitals with the greatest economic, population and health development were the most affected by the coronavirus following one month of recording the first index case in Brazil. That said, restrictive measures of populational mobility, as well as social distancing, could be more effective if directed to capitals with a large flow of people.
The particular characteristics of Macapá delineated in this study is its remoteness from the main national socioeconomic axis not having land routes of communication with other major cities as well as having a reduced number of flights to other cities which has contributed to the lower incidence rate until March 26, 2020. Macapá is the Brazilian capital with the fewest health facilities. This certainly raises questions about asymptomatic carriers as the public health infrastructure in this city faces some limitations. However, due to coordinated efforts among various stakeholders, including the local university, the percent diagnostic rate in Macapá remains higher compared to other Brazilian states with better socioeconomic and health indicators.
To date, geographical factors may contribute to a lower transmission rate of the virus in the Amazon, especially in small to medium urban centers, such as Macapá. These factors, combined with a governmental measure of population social distancing, investments in disease prevention and awareness, as well as scientific research aimed at combating COVID-19 are essential steps to reduce public health system burden of the disease as most of these urban centers are fragile due to historical inequality in capacity-building to confront a pandemic virus.
ACKNOWLEDGMENT
We are grateful for the collaboration of João Silvestre da Silva-Júnior, PhD in Public Health from the Faculty of Public Health of the University of São Paulo and Virgílio Amaral da Cunha Junior, journalist graduated from the Federal University of Viçosa.
REFERENCES
AP. Governo do Estado do Amapá. Decreto n° 1.414 de 19 de Março de 2020. Dispõe sobre medidas de restrição de aglomeração de pessoas com a finalidade de reduzir os riscos de transmissão do novo Coronavírus (COVID-19) e adota outras providências. Macapá, AP: 2020.
ATLAS BRASIL. Atlas do Desenvolvimento Humano no Brasil. Disponível em: < http://atlasbrasil.org.br/2013/pt/consulta/ >. Access in: 24 march 2020.
BRASIL. Ministério da Saúde. Secretaria de Atenção Primária à Saúde. Protocolo de Manejo Clínico do Coronavírus (Covid-19) na Atenção Primária à Saúde. 1. ed. Brasília, DF: Ministério da Saúde – Secretaria de Atenção Primária à Saúde. 2020a.
______. Ministério da Saúde. Secretaria de Vigilância em Saúde. Boletim Epidemiológico 02: Infecção humana pelo Novo Coronavírus (2019-nCov). Brasilia, DF: Ministério da Saúde – Centro de Operações de Emergências em Saúde Pública (COE-nCoV): 2020b.
______. Ministério da Saúde. Secretaria de Atenção Especializada à Saúde. Protocolo de Tratamento do Novo Coronavírus (2019-nCoV). 1. ed. Brasília, DF: Ministério da Saúde – Secretaria de Atenção Especializada à Saúde. 2020c.
_______. Ministério da Saúde. Coronavírus: 25 mortes e 1.546 casos confirmados. Brasilia, DF: 2020d. Disponível em: < https://www.saude.gov.br/noticias/agencia-saude/46573-coronavirus-25-mortes-e-1-546-casos-confirmados >. Access in: 23 March 2020.
______. Ministério da Saúde. Portaria nº 454, de 20 de Março de 2020. Declara, em todo o território nacional, o estado de transmissão comunitária do Coronavírus (Covid-19). Brasília, DF: 2020e. Disponível em: < http://www.in.gov.br/en/web/dou/-/portaria-n-454-de-20-de-marco-de-2020-249091587 >.Access in: 24 March 2020.
_______. Instituto Brasileiro de Geografia e Estatística. IBGE Cidades e Estados. 2020f. Disponível em: < https://www.ibge.gov.br/cidades-e-estados >. Access in: 23 March 2020.
______. Ministério da Saúde. DATASUS. Cadastro Nacional dos Estabelecimentos de Saúde do Brasil – CNES. Quantidade por Tipo de Prestador segundo Capital. Brasília, DF: 2020g. Available at: < https://nam10.safelinks.protection.outlook.com/?url=http%3A%2F%2Ftabnet.datasus.gov.br%2Fcgi%2Ftabcgi.exe%3Fcnes%2Fcnv%2Festabbr.def&data=02%7C01%7C%7C5789bcef877546b7609e08d7d5ac6237%7C84df9e7fe9f640afb435aaaaaaaaaaaa%7C1%7C0%7C637212808662483071&sdata=AZP0PC%2BzxxMGbMTJLdIJO0zLGtXCF9F3BthmFJnvqgo%3D&reserved=0 >. Access in 24 march 2020.
______. Ministério da Saúde. Secretaria de Vigilância em Saúde. Boletim Epidemiológico 05: Doença pelo Coronavírus 2019. Brasilia, DF: Ministério da Saúde – Centro de Operações de Emergências em Saúde Pública (COE-nCoV): 2020h.
______. Ministério da Saúde. Sobre a doença. Brasília, DF: 2020i. Available at: < https://coronavirus.saude.gov.br/sobre-a-doenca>. Access in: 03 april 2020.
______. Agência Nacional de Aviação Civil. Consulta Interativa – Indicadores do Mercado de Transporte Aéreo. Brasília, DF: 2020j. Available at: < https://www.anac.gov.br/assuntos/dados-e-estatisticas/mercado-de-transporte-aereo/consulta-interativa/demanda-e-oferta-origem-destino >. Access in: 23 march 2020
______. Instituto Brasileiro de Geografia e Estatística. Pesquisa Nacional por Amostra de Domicílios Contínua – PNAD Contínua 2016-2018, Características Gerais dos Moradores. Brasília, DF: 2018a. Available at: < https://www.ibge.gov.br/estatisticas/sociais/populacao/17270-pnad-continua.html?edicao=24437&t=downloads >. Access in: 24 March 2020.
______. Ministério da Economia. Relação Anual de Informações Sociais: Amapá 2018. Brasília, DF: 2018b. Available at: < http://pdet.mte.gov.br/rais?view=default >. Acces in: 03 april 2020.
______. Instituto Brasileiro de Geografia e Estatística. Coordenação de População e Indicadores Sociais. Síntese dos indicadores sociais 2018: uma análise das condições de vida da população brasileira. Brasília, DF: 2018c.
______. Instituto Brasileiro de Geografia e Estatística. Amazônia Legal: Municípios da Amazônia Legal. Brasília, DF: 2014. Available at: < https://www.ibge.gov.br/geociencias/informacoes-ambientais/geologia/15819-amazonia-legal.html?=&t=acesso-ao-produto >. Access in: 24 March 2020.
______. Instituto Brasileiro de Geografia e Estatística – IBGE. Atlas de Saneamento – Glossário. Brasília., DF: 2011. Available at: https://biblioteca.ibge.gov.br/visualizacao/livros/liv53096_glossario_equipetec.pdf. Access in: 29 March. 2020.
______. Ministério da Saúde. Guia de Vigilância Epidemiológica. Brasília, DF:. 2009. Available at: < https://bvsms.saude.gov.br/bvs/publicacoes/guia_vigilancia_epidemiologica_7ed.pdf>. Access in: 01 april 2020.
CAMPELLO, T.; GENTILI, P.; RODRIGUES, M.; HOEWELL, G.R. Faces da desigualdade no Brasil: um olhar sobre os que ficam para trás. Saúde em Debate. v.42 n. especial p.54-66, 2018. https://doi.org/10.1590/0103-11042018S305
CASCELLA, M.; RAJNIK, M.; CUOMO, A.; DULEBOHN, S.; DI NAPOLI, R. Features, Evaluation and Treatment Coronavirus (COVID-19). [Atualizado em 8 março 2020] Treasure Island, FL: StatPearls Publishing; 2020. Available at: <https://www.ncbi.nlm.nih.gov/books/NBK554776/ > Access in: 23 March 2020.
DOREMALEN, N.V. et al. [Correspondence]. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. Destinatário: The New England Journal of Medicine. Massachusetts, 17 March, 2020. https://doi.org/10.1056/NEJMc2004973
DRUMMOND, J.A. Investimentos privados, impactos ambientais e qualidade de vida num empreendimento mineral amazônico: o caso da mina de manganês de Serra do Navio (Amapá). História, Ciências, Saúde-Manguinhos. Rio de Janeiro, v. 6, supl. p. 753-792, 2000. https://doi.org/10.1590/S0104-59702000000500002
FAN, J.; LIU, X.; PAN.; DOUGLAS, M.W.; BAO, S. Epidemiology of 2019 Novel Coronavirus Disease-19 in Gansu Province, China, 2020. Emerging Infectious Diseases. v. 26 n.6, 2020a. https://doi.org/10.3201/eid2606.200251
FAN, C. et al. Prediction of Epidemic Spread of the 2019 Novel Coronavirus Driven by Spring Festival Transportation in China: A Population-Based Study. International Jornal of Envirommental Research and Public Health. v.17 n.5, 2020b. https://doi.org/10.3390/ijerph17051679
FARIA, N.R. et al. Genomic and epidemiological monitoring of yellow fever virus transmission potential. Science. v.361 n. 6405 p.894-899, 2018. https://doi.org/10.1126/science.aat7115
GALLASCH, C.H.; CUNHA, M.L; PEREIRA, L.A.S.; SILVA-JUNIOR, J.S. Prevenção relacionada à exposição ocupacional do profissional de saúde no cenário de COVID-19. Revista Enfermagem UERJ. V.28 e.49596. https://doi.org/10.12957/reuerj.2020.49596
GIOVANETTI, M. et al. Genomic and Epidemiological Surveillance of Zika Virus in the Amazon Region. Cell Reports. v.30 n.7, 2020. https://doi.org/10.1016/j.celrep.2020.01.085
HAFFAJEE, R.L.; MELLO, M.M. Thinking Globally, Acting Locally: The U.S. Response to Covid-19. The New England Journal of Medicine, 2020. https://doi.org/10.1056/NEJMp2006740
KAMPF, G.; TODT, D.; PFAENDER, S.; STEINMANN, E. Persistence of coronaviruses on inanimate surfaces and their inactivation with biocidal agents. The Journal of Hospital Infection. v. 104, n. 3, p. 246–251, 2020. https://doi.org/10.1016/j.jhin.2020.01.022
KUCHARSKI, A.J. et al. Early dynamics of transmission and control of COVID-19: a mathematical modelling study. The Lancet: Infectous Diseases. 11 March 2020. https://doi.org/10.1016/S1473-3099(20)30144-4
KOH, D. Occupational risks for COVID-19 infection. Occupational Medicine. v.70 n.1 p. 3-5, 2020. https://doi.org/10.1093/occmed/kqaa036
LAI, S.; BOGOCH, I.I.; WALTS, E.; KHAN, K.; LI, Z.; TATEM, A. Preliminary risk analysis of 2019 novel coronavirus spread within nad beyond China. World pop. In press, 2020. Available at: < https://www.worldpop.org/resources/docs/china/WorldPop-coronavirus-spread-risk-analysis-v2.pdf >. Access in: 02 April 2020.
LEVIN J.; FOX J.A.; FORDE D.R. Elementary statistics in social research. 12ª ed. Upper Saddle River: Pearson Education; 2014.
LEVORATO, C.D.; MELLO, L.M.; SILVA, A.S.; NUNES A.A. Fatores associados à procura por serviços de saúde numa perspectiva relacional de gênero. Ciência e Saúde Coletiva. v. 19 n.4 p. 1263-74, 2020. https://doi.org/10.1590/1413-81232014194.01242013
LIPSITCH, M.; PHIL, D.; SWERDLOW, D.L.; FINELLI, L. Defining the Epidemiology of Covid-19 — Studies Needed. The New England Journal of Medicine. 2020. https://doi.org/10.1056/NEJMp2002125
LI, W. et al. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. v. 426, 2003. https://www.nature.com/articles/nature02145.pdf
LI, Y.C.; BAI W.Z.; HASHIKAWA, T. The neuroinvasive potential of SARS‐CoV2 may be at least partially responsible for the respiratory failure of COVID‐19 patients. Journal of Medical Virology. v. 1-4, 2020. https://doi.org/10.1002/jmv.25728
MAPA DO CORONA VÍRUS. POLATO, A.; CUNHA, R.; SORANO, V. (Coord.). Casos de coronavírus no Brasil e no Mundo: mapa e evolução. Available at: < https://especiais.g1.globo.com/bemestar/coronavirus/mapa-coronavirus/?_ga=2.48817062.1083509909.1584787216-1227695381.1584787197#/.> Access in: 26 march. 2020.
MCINTOSH, K. UpToDate. Coronavirus Disease 2019 (COVID-19). USA: 2020. Available at: < https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19 >. Acesso em: 23 march 2020.
NASSIRI, R. Perspective on Wuhan Viral Pneumonia. Advances in Public Health, Community and Tropical Medicine, V. 02, 2020.
OLIVEIRA, N.M.; PIFFER, M.; STRASSBURG, U. O Indicador de Desenvolvimento Regional no Território do Tocantins. Interações. v.20 n.1 p. 3-20, 2019. http://dx.doi.org/10.20435/inter.v0i0.1607
OLIVEIROS, B.; CARAMELO, L.; FERREIRA, N.C.; CARAMELO, F. Role of temperature and humidity in the modulation of the doubling time ofCOVID-19 cases. Medrxiv. 2020. https://doi.org/10.1101/2020.03.05.20031872
PREM, K. et al. The effect of control strategies to reduce social mixing on outcomes of the COVID-19 epidemic in Wuhan, China: a modelling study. The Lancet. 25 March 2020.https://doi.org/10.1016/S2468-2667(20)30073-6
READ, J.M.; BRIDGEN, J.R.E.; CUMMINGS, D.A.T.; HO, A.; JEWELL, C.P. Novel coronavirus 2019-nCoV: early estimation of epidemiological parameters and epidemic predictions. Medrxiv. In press, 2020. https://doi.org/10.1101/2020.01.23.20018549
RIPAP. Rede Integrada de Pesquisa do Amapá. Carta Manifesto da Comunidade Científica Amapaense. Macapá. 2020. Available at: < https://drive.google.com/file/d/1Kgsikv5B27v60QPyRR6gpyEVjmnVOVfF/view >. Access in : 03 april 2020.
ROSA, S.J. Transporte e Exclusão Social: A Mobilidade da População de Baixa Renda da Região Metropolitana de São Paulo e Trem Metropolitano. 2006. Dissertação (Mestrado em Engenharia) – Escola Politécnica da Universidade de São Paulo, São Paulo, 2006. Available at: < https://www.teses.usp.br/teses/disponiveis/3/3138/tde-07122006-163515/publico/dissertacao_silvio_jose_rosa.pdf >. Access in: 04 april 2020.
ROTHAN, H.A.; BYRAREDDY, S.N. The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak. Journal of Autoimmunity. In press, 2020. https://doi.org/10.1016/j.jaut.2020.102433
ROTHE, C. et al., [Correspondece]. Transmission of 2019-nCoV Infection from an Asymptomatic Contact in Germany. Destinatário: The New England Journal of Medicine. Massachusetts, 5 March, 2020. https://doi.org/10.1056/NEJMc2001468
SESACE. Governo do Estado do Ceará. Secretaria de Saúde. Boletim epidemiológico: Doença pelo novo coronavírus (COVID-19) n° 18. Fortaleza, CE: 2020.
SESAMA. Governo do Estado do MaranhãO. Secretaria de Saúde. Nota 9 – SES monitora 205 casos suspeitos de COVID-19. São Luís, MA: 2020.
SESAMT. Governo do Estado do Mato Grosso. Secretaria de Saúde. Secretaria Adjunta de Atenção e Vigilância em Saúde. Nota Informativa 16 COVID-19. Cuiabá, MT: 2020.
SESAMG. Governo do Estado de Minas Gerais. Secretaria de Estado de Saúde de Minas Gerais. Subsecretaria de Vigilância em Saúde. Centro de Operações de Emergência em Saúde Pública. Boletim Informativo Diário de 20/03/2020. Belo Horizonte, MG: 2020a.
SESAMG. Governo do Estado de Minas Gerais. Secretaria de Estado de Saúde de Minas Gerais. Subsecretaria de Vigilância em Saúde. Centro de Operações de Emergência em Saúde Pública. Boletim Informativo Diário de 14/03/2020. Belo Horizonte, MG: 2020b.
SESAMG. Governo do Estado de Minas Gerais. Secretaria de Estado de Saúde de Minas Gerais. Subsecretaria de Vigilância em Saúde. Centro de Operações de Emergência em Saúde Pública. Informe Epidemiológico n°3, de 03 de Março de 2020. Belo Horizonte, MG: 2020c.
SESARS. Governo do Estado do Rio Grande do Sul. Secretaria de Saúde. Centro Operações de Emergências Rio Grande do Sul. Informe Epidemiológico – COVID-19 EM 19/03/2020. Porto Alegre, RS: 2020.
SILVA, D.R.; VIANA, V.P.; MÜLLER, A.M.; LIVI, F.P.; DALCIN, P.T.R. Respiratory viral infections and effects of meteorological parameters and air pollution in adults with respiratory symptoms admitted to the emergency room. Influenza and others Respiratory Viruses. v. 8 n. 1, 2013. https://doi.org/10.1111/irv.12158
TORMAN, V.B.L.; COSTER, R.; RIBOLDI, J. Normalidade de variáveis: métodos de verificação e comparação de alguns testes não-paramétricos por simulação. Revista HCPA. Porto Alegre, v. 32, n. 2, p. 227-234, 2012.
TRAVASSOS, C.; OLIVEIRA, E.X.G.; VIACAVA, F. Desigualdades geográficas e sociais no acesso aos serviços de saúde no Brasil : 1998 e 2005. Ciência & Saúde Coletiva. v.11 n.4 p.975-986, 2006. https://doi.org/10.1590/S1413-81232006000400019
VELAVAN, T.P.; MEYER, C.G. The COVID‐19 epidemic. Tropical Medicine & International Health. v. 25, n.3, 2020. https://doi.org/10.1111/tmi.13383
VIANA, A.L.D.; IBAÑEZ, N.; ELIAS, P. E. M. Saúde, desenvolvimento e território. 1 ed. São Paulo: Hucitec; 2009.
WANG, J.; TANG, K.; FENG, K.; LV.W. High Temperature and High Humidity Reduce the Transmission of COVID-19. SSRN. 2020. https://dx.doi.org/10.2139/ssrn.3551767
WHO. Word Health Organization. Report of the WHO-China Joint Mission on Coronavirus Disease 2019 (COVID-19). China: Word Health Organization – The Joint Mission. 2020a. Available at: < https://www.who.int/docs/default-source/coronaviruse/who-china-joint-mission-on-covid-19—final-report-1100hr-28feb2020-11mar-update.pdf?sfvrsn=1a13fda0_2&download=true >
WHO. World Health Organization. Coronavirus disease 2019 (COVID-19): Situacional Report – 62. Genebra, Switzerland. WHO: 2020b.br
WHO. World Health Organization. Coronavirus disease 2019 (COVID-19): Situacional Report – 38. Genebra, Switzerland. WHO: 2020c.
WU, J.T.; LEUNG, K.; LEUNG, G.M.; Nowcasting and forecasting the potential domestic and international spread of the 2019-nCoV outbreak originating in Wuhan, China: a modelling study. The Lancet. v.395 n.10225 p.689-697, 2020. https://doi.org/10.1016/S0140-6736(20)30260-9
ZASLAVSKY, R.; GOULART, B. N. G. Migração pendular e atenção à saúde na região de fronteira. Ciência e Saúde Coletiva. 2017, vol.22, n.12. https://doi.org/10.1590/1413-812320172212.03522016
ZHANG, Y. et al. The Novel Coronavirus Pneumonia Emergency Response Epidemiology Team. The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. China CDC Weekly. v. 2, n. x, 2020. https://doi.org/10.3760/cma.j.issn.0254-6450.2020.02.003
[1] Physician, Specialist in Health Systems and Services Management. Professor, preceptor and researcher of the Medicine Course at Campus Macapá, Federal University of Amapá (UNIFAP).
[2] Medical Student at the Macapá Campus, Federal University of Amapá (UNIFAP).
[3] Medical Student at the Macapá Campus, Federal University of Amapá (UNIFAP).
[4] Medical Student at the Macapá Campus, Federal University of Amapá (UNIFAP).
[5] Biologist, PhD in Theory and Research of Behavior, Professor and researcher of the Chemistry Degree Course at the Basic, Technical and Technological Institute of Amapá (IFAP).
[6] Physician, D.sc in Hematology, Fellowship in Clinical Pharmacology, expertise in Global Health and Viral Infections. Professor, Department of Pharmacology and Toxicology and, Family and Community Medicine, Michigan State University (MSU), Michigan, USA.
[7] Nurse. Health Secretary of the Municipality of Macapá AP (SESA AP).
[8] Nurse. Master in Health Sciences. Amapá Municipal Health Secretariat (SESA AP).
[9] Nurse. PhD in Sciences. Amapá Municipal Health Secretariat (SESA AP).
[10] Nurse. Amapá Municipal Health Secretariat (SESA AP).
[11] Physician, Master in Teaching and Health Sciences, Professor, preceptor and researcher of the Medical Course at Campus Macapá, Federal University of Amapá (UNIFAP).
[12] Biologist, PhD in Topical Diseases, Professor and researcher at the Physical Education Course at, Federal University of Pará (UFPA).
[13] Theologian, PhD in Psychoanalysis, researcher at the Center for Research and Advanced Studies – CEPA.
[14] Biomedical, PhD in Topical Diseases, Professor and researcher of the Medicine Course at Campus Macapá, Federal University of Amapá (UNIFAP).
Enviado: Abril, 2020.
Aprovado: Abril, 2020.