ORIGINAL ARTICLE
BANDEIRA, Debora Marina [1], MARCHI, Juliana Pelissari [2], GOMES, Simone Damasceno [3], PELLIZZARO, Luciana [4]
BANDEIRA, Debora Marina. et al. Antimicrobial activity of Curcuma longa L. on Escherichia coli. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year. 08, Ed. 04, Vol. 03, pp. 05-16. April 2023. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/biology/antimicrobial-activity, DOI: 10.32749/nucleodoconhecimento.com.br/biology/antimicrobial-activity
ABSTRACT
Curcuma longa is a plant that has stood out in research evaluating antimicrobial, anti-inflammatory, antiprotozoal, anticarcinogenic and antifungal activity. The discovery of new alternatives for the development of drugs originating from active principles of plants is necessary considering the high adaptive capacity of different microorganisms. Thus, the present study aimed to evaluate the antimicrobial activity of extracts of oleoresin and powder suspension of rhizomes of Curcuma longa against the bacterium Escherichia coli. In this study, no antimicrobial activity was observed for the extracts of turmeric powder and oleoresin against Escherichia coli. There is a wide variety of results obtained in the literature in the face tests of C. longa on the different microorganisms, some positive, demonstrating antimicrobial action, and others negative. This divergence is justified by factors such as: different methodologies used, climate, soil types, fertilization, water availability of the plant’s cultivation, as well as the form of harvest and storage, which interfere in the components present and in their concentration in the plant. Although the results of this study were negative, it does not mean that the plant does not have antimicrobial activity, being indicated the continuity in the assays, aiming to identify the factors that generate disagreement between the studies already carried out.
Keywords: Natural products, Oil resin, Turmeric powder, Curcumin, Antimicrobial activity.
1. INTRODUCTION
Natural products have been used throughout history in the treatment and prevention of diseases. Currently, they have been gaining great importance in research for the development of drugs of natural origin. Several groups of researchers have studied the biological activity of medicinal plants originating in various parts of the world, guided by the popular use of native species (SILVA FILHO et al., 2009; SANTOS et al., 2021; BANDEIRA et al., 2022).
A plant that, in addition to being used in cooking, has been present in research in the pharmacological areas is Curcuma longa L., popularly known as turmeric or turmeric. It is an herbaceous plant, belongs to the Zingiberaceae family, native to Southeast Asia, more precisely to the tropical forests of India, the country with the largest production in the world (PEREIRA; Stringheta, 1998; FRANCO et al., 2007; SABIR et al., 2021).
The part of the vegetable with greater use is the rhizome, in which are found the substances responsible for its market value and therapeutic action (COLLINO, 2014). There are three curcuminoid pigments present in the rhizome: curcumin (the main compound responsible for the various activities of the plant), demethoxycurcumin and bisdemethoxycurcumin (SILVA FILHO et al., 2009; SABIR et al., 2021). Pereira and Stringheta (1998), explain that turmeric has three commercially available products: turmeric powder, turmeric oleoresin and curcumin extract.
Research with C. longa proved its antimicrobial activity (MAIA et al., 2003; KALAYCIOGLU et al., 2017), anti-inflammatory, antioxidant, antiprotozoal, anticarcinogenic and antifungal (AKRAM et al., 2010; MARCHI et al., 2016). Faced with the resistance that pathogenic microorganisms have developed, there is a need and encouragement for the search for drugs of natural origin (BANDEIRA et al., 2022).
The bacterium E. coli, for example, is a commensal microorganism, present in the intestines of mammals and birds, which has been pointed out as one of the most frequent bacterial agents in diarrhea of humans and animals, and can cause, in humans, several infections, including meningitis (FERREIRA; KNÖBL, 2009; ALMEIDA, 2013).
The high adaptive capacity of microorganisms makes research on their behavior essential for public health. The World Health Organization (WHO) recently released the first list of antibiotic-resistant “priority pathogens,” with 12 families of bacteria posing the greatest threat to human health. In this list, the threat of gram-negative bacteria, resistant to multiple antibiotics, such as, stands out E. coli, which along with other Enterobacteriaceae, are on the list as priority 1, that is, “critical, resistant to carbapenem and third-generation cephalosporins, the best antibiotics available for the treatment of multidrug-resistant bacteria” (WHO, 2018). Also, E. coli has between 8% and 65% (variable between different countries) of resistance to ciprofloxacin, an antibiotic commonly used to treat urinary tract infections (WHO, 2017).
Thus, it is necessary to discover new alternatives for the development of drugs originating from the active principles of some plants. In this context, this study aimed to evaluate the antimicrobial activity of oleoresin extract and powder suspension of rhizomes C. longa on the bacterium E. coli, in view of being products already available to consumers.
2. MATERIAL AND METHODS
The study was carried out at the Microbiology Laboratory of the Universidade Paranaense, University Unit of Francisco Beltrão, and the experimental part was carried out from May to October 2018.
The rhizomes of Curcuma longa were acquired in the trade of Francisco Beltrão, Paraná. They were washed, sanitized in running water, peeled, sliced to a thickness of approximately 0.5 cm. After that, the slices were dried in an oven at 50 ºC, until the verification of constant mass. The total drying period was approximately one month. The constant mass weight was 516.63 g.
After drying, the powder was obtained by grinding the rhizomes in a domestic blender, followed by sieving through four meshes of different thicknesses. The powder was packed in double plastic bags (one layer of kraft paper and another of plastic bag, until its use) and kept in a dry environment (MARCHI, 2018).
The turmeric rhizome powder was divided into: 180 g to be made the alcoholic extract to obtain the oleoresin (OR); and 173.24 g to obtain the powder suspension (PS).
To obtain the OR, it was started by making a crude extract (CE), being used the maceration process with 70% ethyl alcohol at room temperature (PEREIRA; Stringheta, 1998; RODRIGUES et al., 2016). We used 180 g of turmeric powder for 540 ml of 70% alcohol (1:3 (m/v) plant/solvent); The material was homogenized with the aid of a glass stick, filled in a glass container surrounded by aluminum foil, labeled for identification (name, date, time) and stored in a dark place for 15 days, with daily agitation.
After fifteen days, the CE was filtered in vacuum. For this, a Buchner funnel was coupled to a kitassate with rubber, for complete sealing, and from the kitassate followed a hose to the vacuum pump. After assembling the equipment, the funnel received two filter papers in the form of a circle, in which the CE was deposited. Then, the device was turned on and the CE was suctioned by the pump, allowing its filtration. The filtrate was directed to the interior of the kitassate, obtaining the tincture, containing alcohol, totaling 350 ml. The solid material that was retained in the filter paper was later discarded (MARCHI, 2018).
The tincture obtained was placed in a rotaevaporator to remove the ethyl alcohol. To this end, the tincture was placed in a flat-bottomed balloon, which was connected to the rotaevaporator and heated in a water bath at a temperature of 45 + 5 ºC.
The procedure was performed until 1/5 of the tincture was in the balloon, and from then on, it was called OR (20%). The material was packed in falcon tubes, wrapped in aluminum foil and stored in a refrigerator until dilution for the assembly of the experiment (MARCHI, 2018).
Regarding the OR, the pure preparation obtained after rotaevaporation was considered as 100% and is diluted in concentrations of 50; 25; 12.5 and 6.25%, in Tween 80 (at 0.1%). To prepare the suspension of the powder at 20%, the addition of 1.875 g of powder was made in 7.5 ml of a sterile solution of Tween 80 (0.1%), according to the methodology used by Dornellas (2016).
Strains of Escherichia coli were acquired and reconstituted according to the supplier’s indication. Microbial cultures were standardized at 108 cells/ml, estimated by comparison to the 0.5 tube of the McFarland scale.
For the assay, we adopted the Agar Plug Diffusion Method, adapted from Kurtzman Jr (1973) and Harris and Ruger (1953), which consists of removing a piece of the culture medium from the site corresponding to the repique after the incubation period and placing them on the surface of the culture medium inoculated with the material to be tested, where, after that, growth inhibition will be observed due to the diffusion of the product to be tested and/or the antibiotic from the plug. Thus, Petri dishes of 100 x 20 mm in diameter were used, in which 20.0 ml of Muller Hinton agar prepared as indicated on the package were added. After being homogenized and left to rest until solidification with a sterile swab, the culture of E. coli was inoculated uniformly over the surface of the agar plates, which were left to rest at room temperature for approximately three minutes.
In parallel, the plates containing 15.0 ml of sterile medium, plus 5.0 ml of powder suspension or OR previously diluted in their concentrations (3:1 medium/extract) were prepared. The mixture was homogenized and awaited for solidification. Then, the cylindrical plugs (cutouts) of each of these plates were removed to the medium containing the inoculum. In each plate, five plugs were transferred in their proper concentrations – from the highest to the lowest OR (100%, 50, 25, 12.5 and 6.25%) – as well as plugs with positive control – using amoxicillin and chloramphenicol, and with negative control, using Tween 80 to 0.1%.
The experiment was carried out in quintuplicate and the plates incubated at 40 + 3 ºC, for 24 hours (MARTINS, 2010; MINAGAWA, 2007).
For the analysis of the PS, the same procedure was followed, but adding to the plates with culture medium and innocuous, plugs containing the PS at 20%, in the same concentrations of the OR (100; 50; 25; 12.5 and 6.25%).
After the interval of 24 hours, the measurement of the inhibition halos, when formed, was performed with a caliper, according to the criteria recommended by the National Committee for Clinical Laboratory Standards (NCCLS, 2003).
3. RESULTS AND DISCUSSION
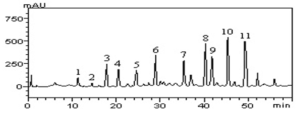
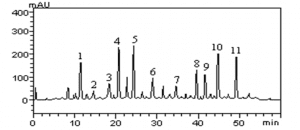
In this study, no antimicrobial activity was observed for the extracts of the suspension of turmeric powder (PS) and oleoresin (OR) against the bacterium Escherichia coli, in any of the tested concentrations. (Figure 1).
Figure 1: Comparison of the presence or absence of inhibition halos
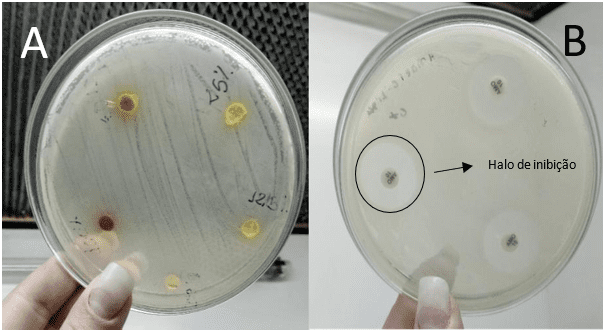
This non-inhibition of E. coli by turmeric products has been tested in other studies. Péret-Almeida (2006), for example, evaluated the antimicrobial activity of turmeric powder, curcuminoid pigments and turmeric essential oil. In this experiment, only the essential oil had antimicrobial activity on E. coli, Bacillus subitilis, Salmonella enterica Choleraesuis, Aspergillus niger and Saccharomyces cereviseae. The same was observed in the studies of Norajit et al. (2007), Araújo et al. (2015) and Franco et al. (2007), which also tested turmeric products and there was no activity against E. coli. The non-action of C. longa v.s. E. coli can be justified by peculiarities of the bacterium or plant.
As for E. coli, as well as other bacteria, it is believed that the pathogenic and resistant potential of some strains is due to genetic gains occurred during the evolutionary process of the species, due to the acquisition of virulence genes, through mutations or horizontal transfer of genetic material, which made them develop complex mechanisms of resistance (VIEIRA, 2009). These virulence genes, coming from extrachromosomal genetic materials (plasmids), encode proteins that enable colonization, penetration and invasion of new spaces by some bacteria (CORRÊA, 2012), making it difficult to control.
Similarly, the antimicrobial activity for gram-negative bacteria is lower when exposed to some antimicrobials, associating this fact with the presence of the outer membrane in the structure of the bacteria, which can restrict the penetration of certain substances (SINGH et al., 2002; GUL et al., 2004).
Despite this, free curcumin – pigment present in turmeric – and curcumin microcrystals showed antimicrobial activity on E. coli, Staphylococcus aureus, Bacillus cereus and Pseudomonas aeruginosa (SANTOS, 2015; DORNELLAS, 2016). Curcumin was also effective in another study, against gram-negative bacteria such as E. coli and S. chorelaesuis (TAKEUCHI, 2012). The ethanol extract and essential oil of C. longa also acted against standard strains and clinical isolates of E. coli, with the ethanol extract causing greater inhibition compared to the oil (ÀLVAREZ et al., 2016).
In this context, it is possible to realize that ethanol extract and curcumin were more effective against E. coli. This can be explained by the concentration of curcumin that varies in the rhizomes from one plant to another, as Pereira and Stringheta (1998) pointed out an average curcumin concentration of 2.5% and, Chassagnez; Corrêa and Meireles (1997), 6.4%. The difference in concentration is often associated with the composition of the rhizomes or the way it is extracted.
Thus, the turmeric species evaluated here may have presented low curcumin concentration, influencing the non-antimicrobial activity. Considering that the phytochemical analysis for C. longa used in this essay, this hypothesis cannot be confirmed. Regarding the use of OR, it is intended to concentrate the curcuminoid compounds in relation to the rhizome of the ground plant in powder, because it eliminates the starch present in greater quantity in the rhizome (25 to 50 %) (NAGHETINI, 2006; SABIR et al., 2021). Thus, there is a possibility that OR is more effective in its antimicrobial action, but in this study this was not confirmed.
There are several factors that can influence the concentration of active components in turmeric products. The extraction temperature and particle size have a significant influence on the yield of curcumin extraction with ethanol, according to a study by Peron et al. (2010) with turmeric OR. The drying temperature at 70 ºC favored the extraction of OR, as well as contributed to the maintenance of curcumin in the raw material (CHASSAGNEZ; CORRÊA and MEIRELES, 1997). The drying and rotation temperature of the OR of this study ranged from 40ºC to 50ºC, which may have influenced the curcumin concentrations, but this possibility could only be proven, once again, by phytochemical analyses.
Soil and growing conditions also interfere with the curcumin content of the rhizomes. Peron et al. (2010), proved that there is a significant difference of 95% between the rhizomes grown in Piracicaba compared to the rhizomes grown in Monte Alegre do Sul, both cities in the state of São Paulo.
In addition, there is the extraction method used, which interferes with the physicochemical differences of plant products. Naghetini (2006), observed that the extraction with hexane was simpler, faster and of higher yield when compared to the hydrodistillation. Pereira and Stringheta (1998), on the other hand, state that ethyl alcohol and acetone are indicated as good solvents, with acetone being the most used solvent, even presenting some flammability problems and high cost for its recovery, in order to rid the product of its presence.
A study by Krishnamurthy et al. (1976) on curcuminoid extraction processes with acetone, ethanol and ethylene dichloro concluded that acetone was more efficient than ethanol and ethylene dichlorine, providing a product containing 42% curcuminoids. Thus, indicating that ethanol, the solvent used in this study, may not be as effective in extracting the compounds necessary for antimicrobial action.
4. CONCLUSION
The powdered and OR extracts produced from Curcuma longa marketed in Francisco Beltrão, Paraná, did not show antimicrobial activity for Escherichia coli strains.
This result may be linked to several factors, such as: the possible resistance of the bacteria to plant compounds or environmental and management conditions, such as: temperature, soil type, fertilization, water availability, harvest time and storage time, which may influence the chemical composition and concentration of PS and OR components, that have been effective in other studies, such as curcumin.
It is suggested a continuity in the trials, aiming to identify which or what are the factors that cause disagreements with the literature, as well as the analysis of the components present in the tested products.
5. ACKNOWLEDGMENT
We thank the Universidade Paranaense for the study environment provided; the Guiding Professor Luciana Pellizzaro, for all the time invested in carrying out this work; the Co-advisor professor, Juliana Pelissari Marchi, for her experience in the subject and assistance in carrying out this work; and Professor Simone for her encouragement in publishing my work.
REFERENCES
AKRAM, M.; UDDIN, S.; AHMED, F.; USMANGHANI, K.; HANNAN, A. MOHIUDDIN, E.; ASIF, M. Curcuma longa and Curcumin: A review article. Romanian Journal Biology – Plant Biology, Bucharest, vol. 55, n. 2, p. 65-70, 2010.
ALMEIDA, A. M. S. Características biológicas e antigênicas de Escherichia coli com ênfase aos genes de virulência. Seminário (Pós-Graduação em Ciência Animal) – Universidade Federal de Goiás, Goiânia, 2013. 30 f.
ÀLVAREZ, N. M.; ORTÍZ, A. A.; MARTÍNEZ, C. O. Atividad antibacteriana in vitro de Curcuma longa (Zingiberaceae) frente a bactérias nosocomiales em Montería, Colombia. Revista Biologia Tropical, vol. 64, n. 3, p. 1201-1208, 2016.
ARAÚJO, R. G. M.; ASSIS, D.; LEMES, S. R.; MELO-REIS, P. R; ARAÚJO, L. A.; PAIVA, E. S.; SILVA, C. B. Avaliação da atividade antimicrobiana do óleo essencial do açafrão (Curcuma longa): estudo de caso. PUC, Goiânia, vol. 42, n. 4, p. 425-431, 2015.
BANDEIRA, D. M.; CORRÊA, J. M.; LASKOSKI, L. V.; BATISTA, J. M.; ROSSET, J.; COSTA, W. F.; KUO, L. H.; PINTO, F. G. S. Extraction, characterization of bioactive compounds and biological activities of the leaves of Podocarpus lambertii Klotzch ex Endl. J Appl Res Med Aromat Plantas, n. 31, p. 100427, 2022.
CHASSAGNEZ, A. L. M.; CORRÊA, N. C. F.; MEIRELES, M. A. A. Extração de oleoresina de cúrcuma (Curcuma longa L.) com CO² surpercrítico. Ciênc. e Tecnol. de Aliment., Campinas, vol. 17, n. 4, 1997.
COLLINO, L. Curcumina: de especiaria a nutracêutico. Trabalho de Conclusão de Curso (Graduação em Farmacêutica – Bioquímica), Universidade Estadual Paulista. Araraquara, 2014. 88f.
CORRÊA, F. A. F. Características dos patótipos de E. coli e implicações de E. coli patogênica para aves em achados de abatedouros frigoríficos. Seminário (Pós- Graduação em Ciência Animal) – Universidade Federal de Goiás, Goiânia, 2012. 37 f.
DORNELLAS, F. C. Atividade antifúngica de Curcuma longa L. (Zingiberaceae) contra fungos deteriorantes de pães. Trabalho de Conclusão de Curso (Graduação em Tecnologia em Alimentos) – Universidade Tecnológica Federal do Paraná. Campo Mourão, 2016. 39 f.
FERREIRA, A. J. P.; KNÖBL, T. Enfermidades bacterianas. In: JÚNIOR BERCHIERI, A. et al. Doenças das aves. 2ª Ed. Campinas: Facta, 2009. pp. 457-474.
FRANCO, A. L. P.; OLIVEIRA, T. B.; FERRI, P. H.; BARRA, M. T.; PAULA, J. R. Avaliação da composição química e atividade antibacteriana dos óleos essenciais de Aloysia gratíssima (Gillies & Hook) Tronc. (alfazema), Ocimum gratissimum L. (alfavaca-carvo) e Curcuma longa L. (açafrão). Rev. Eletrônica de Farmácia, vol. 5, n. 2, p. 228-88, 2007.
GUL, N.; AHMAD, S.; MUJAHID, Y. T. Studies on the Antibacterial Effect of Different Fractions of Curcuma longa against urinary tract infection isolantes. Pak J Biol Sci, vol. 7, n. 12, p. 2055-60, 2004.
HARRIS, D. A.; RUGER, M. L. Microbiological aspects of new antibiotic screening. A plug test procedure. Antibiotics & Chemotherapy, vol. 3, n. 1, p. 265-70, 1953.
KALAYCIOGLU, Z.; TORLAK, E.; AKIN-EVINGÜR, G.; ÖZEN, İ.; ERIM, F.B. Antimicrobial and physical properties of chitosan films incorporated with turmeric extract. International Journal of Biological Macromolecules, vol. 101, n. 1, p. 882-888, 2017.
KRISHNAMURTHY, N.; MATHEW, A. G.; NAMBUDIRI, E. S.; SHIVASANKAR, S.; LEWIS, Y. S.; NATARAJAN, C. P. Oil and oleoresin of turmeric. Tropical Science, vol. 18, n. 37, p. 37-45, 1976.
KURTZMAN JR., R. H. Agar-Plug cutter and inoculation device. Mycologia, vol. 65, n. 1, p. 236–39, 1973.
MAIA, S. R.; FERREIRA, A. C.; ABREU, L. R. Uso do açafrão (Curcuma longa L.) na redução da Escherichia coli (ATCC 25992) e Enterobacter aerogenes (ATCC 13048) em ricota. Lavras: UFLA, 2003.
MARCHI, J. P.; TEDESCO, L.; MELO, A. C.; FRASSON, A. C.; FRANÇA, A. C.; SATO, S. W.; LOVATO, E. C. W. Curcuma longa L.: o açafrão-da-terra e seus benefícios medicinais. Arquivo de Ciências da Saúde UNIPAR, Umuarama, vol. 20, n. 3, p. 189-94, 2016.
MARCHI, J. P. Composição química e atividade antioxidante de duas preparações de extrato de rizomas de Curcuma longa L. Dissertação (Mestrado Profissional em Plantas Medicinais e Fitoterápicos na Atenção Básica da Universidade Paranaense) – Universidade Paranaense. Umuarama, 2018.
MARTINS, A. L. G. A. Atividade antibacteriana dos óleos essenciais do manjeirão (Ocimum basilicum Linnaeus) e do gengibre (Zingiber officinale Roscoe) frente a linhagens de Escherichia coli enteropatogênicas isoladas de hortaliças. Tese (Doutorado em Ciência e Tecnologia de Alimentos) – Universidade Federal da Paraíba. João Pessoa, 2010. 110 f.
MINAGAWA, C. W. Estudo microbiológico fecal de linhagens de camundongos, de estirpe de E. coli e do meio ambiente em biotérios. Dissertação (Mestrado em Medicina Veterinária) – Universidade de São Paulo. São Paulo, 2007. 108 f.
NAGHETINI, C. C. Caracterização físico-química e atividade antifúngica dos óleos essenciais da cúrcuma. Dissertação (Mestrado em Ciências de Alimentos) – Universidade Federal de Minas Gerais, Belo Horizonte, 2006.
NATIONAL COMMITTEE FOR CLINICAL LABORATORY STANDARDS – NCCLS. Performance standards for antimicrobial susceptibility testing; tenth informational suplemente. M100-S10. Wayne, 2003.
NORAJIT, K.; LAOHAKUNJIT, N.; KERDCHOECHUEN, O. Antibacterial effect of five Zingiberaceae essential oils. Molecules, vol. 12, n. 8, p. 2047-60, 2007.
ORGANIZAÇÃO MUNDIAL DA SAÚDE – OMS. Novos dados revelam níveis elevados de resistência aos antibióticos em todo o mundo. OMS, 2018. Disponível em: https://www.paho.org/pt/noticias/29-1-2018-novos-dados-revelam-niveis-elevados-resistencia-aos-antibioticos-em-todo-mundo#:~:text=29%20de%20janeiro%20de%202018,de%20alta%20e%20baixa%20renda. Acesso em: 16 abr. 2018.
ORGANIZAÇÃO MUNDIAL DA SAÚDE – OMS. OMS publica de bactérias para as quais se necessitam novos antibióticos urgentemente. OMS, 2017. Disponível em: https://www.paho.org/pt/noticias/27-2-2017-oms-publica-lista-bacterias-para-quais-se-necessitam-novos-antibioticos. Acesso em: 16 abr. 2018.
PEREIRA, A. S.; STRINGHETA, P. C. Considerações sobre a cultura e processamento do açafrão. Hortic. Bras., Brasília, vol. 16, n. 2, p. 102-5, 1998.
PÉRET-ALMEIDA, L. Caracterização de pigmentos da Curcuma longa L., avaliação da atividade antimicrobiana, morfogênese in vitro na produção de curcuminoides e óleos essenciais. Tese (Doutorado em Ciência de Alimentos) – Faculdade de Farmácia, Universidade Federal de Minas Gerais. Belo Horizonte, 2006. 120 f.
PERON, I. L.; CARVALHO, P. R. N.; DA SILVA, M. G.; AZEVEDO FILHO, J. A. De; PINHEIRO, J. B; SIGRIST, M. S.; ZUCCHI, M. I. Avaliação dos teores de curcumina em diferentes acessos de Cúrcuma (Curcuma longa L.). 2010, Anais. Campinas: Escola Superior de Agricultura Luiz de Queiroz, Universidade de São Paulo, 2010.
RODRIGUES, R. R.; GONZÁLEZ, M. E. L.; QUEIROZ, A. A. A. de. Eletrossíntese e caracterização de poli (2-hidroxietilmetacrilato) contendo curcuminoides. Dissertação (Mestrado em Materiais para Engenharia) – Universidade Federal de Itajubá. Itajubá, 2016. 90 f.
SABIR, S. M.; ZEB, A.; MAHMOOD, M.; ABBAS, S.R.; AHMAD, Z.; IQBAL, N. Phytochemical analysis and biological activities of ethanolic extract of Curcuma longa rhizome. Brazilian Journal of Biology – Revista Brasileira de Biologia, vol. 81, n. 3, p. 737-740, 2021.
SANTOS, F. D. P. Avaliação da atividade antimicrobiana de microcristais de curcumina. Trabalho de Conclusão de Curso (Graduação em Engenharia de Alimentos) – Universidade Tecnológica Federal do Paraná. Campo Mourão, 2015. 36 f.
SANTOS, V. C.; MALLMANN, A. P.; TOLEDO, G. A.; BANDEIRA, D. M.; CONCEIÇÃO, L. H. S. M.; CORRÊA, J. M.; PINTO, F. G. S. Phytochemical prospection, antioxidant and antimicrobial activities of leaves extracts from Myrcia palustris DC. Int J Dev Res., n. 11, p. 44724-44729, 2021.
SINGH, G.; SINGH, O. P.; MAURYA, S. Chemical and biocidal ivestigations on essential oils of some Indian Curcuma species. Progress in crystal growth and charecterization of materials, vol. 45, n. 1, p. 75-81, 2002.
SILVA FILHO, C. R. M.; SOUZA, A. G.; CONCEIÇÃO, M. M.; SILVA, T. G.; SILVA, T. M. S.; RIBEIRO, A. P. L. Avaliação da bioatividade de extratos cúrcuma (Curcuma longa L., Zingiberaceae) em Artemia salina e Biomphalaria glabrata. Ver. Bras. de Farmacogn., São Paulo, vol. 19, n. 4, p. 919-23, 2009.
TAKEUCHI, A. P. Caracterização antimicrobiana de componentes do açafrão (Curcuma Longa L.) e elaboração de filmes ativos com montimorilonita e óleo resina de açafrão. Dissertação (Mestrado). Programa de Pós-Graduação em Ciências e Tecnologia de Alimentos – Universidade Federal de Goiás. Goiânia, 2012. 59 f.
VIEIRA, M. A. M. Ilhas de patogenicidade. O mundo da saúde, São Paulo, vol. 33, n. 4, p. 406- 14, 2009.
[1] Master in Conservation and Management of Natural Resources. ORCID: 0000-0001-5956-7210. Curriculum Lattes: https://lattes.cnpq.br/5619420533247630.
[2] Master in Medicinal Plants and Phytotherapics in Primary Care. ORCID: 0000-0002-7779-0253. Lattes Curriculum: http://lattes.cnpq.br/3593944907268063.
[3] PhD in Agronomy. ORCID: 0000-0001-7639-8500. CURRICULUM LATTES: http://lattes.cnpq.br/3362104483832351.
[4] Advisor. Master in Environmental Sciences. ORCID: 0000-0003-1701-9763. CURRICULUM LATTES: http://lattes.cnpq.br/9146011936057512.
Sent: February 23, 2023.
Approved: March 10, 2023.