ORIGINAL ARTICLE
SAVIATO, Mário Junior [1], SASSI, Vitor Bonamigo [2], CUNHA, Mariana Moraes [3], SASSI, Elke Adriana Bonamigo [4], GUIMARÃES JUNIOR, José Carlos [5], LIMA, Jucivaldo Dias [6]
SAVIATO, Mário Junior. Et al. Hematology of Astyanax novae Eigenmann, 1911 (Characidae: Stethaprioninae) in Neblina stream, eastern Amazon, Brazil. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year. 06, Ed. 11, Vol. 15, pp. 198-215. November of 2021. ISSN: 2448-0959, Acess Link: https://www.nucleodoconhecimento.com.br/biology/hematology-of-astyanax, DOI: 10.32749/nucleodoconhecimento.com.br/biology/hematology-of-astyanax
ABSTRACT
This study aimed to identify and measure the hematological parameters (erythrogram, thrombogram, white blood cell count and plasma metabolites) of the species Astyanax novae Eigenmann, 1911, collected in the urban stream, Neblina river in the city of Araguaína – TO, Brazil. The captured animals totaled 60 individuals, which were collected with a cast net and with the aid of a rod and reel with artificial bait. Blood collections were performed by caudal puncture of the caudal blood vessels, and blood data were collected according to the methodology described in the bibliography. From the results of the hematological analysis, mean values were obtained, which were: Hematocrit (Ht): 12.0%; Hemoglobin (Hb): 3.6 g/dL and erythrocytes: (GV): 0.59 x 106/uL. The allusive of variations in these parameters may suggest indicators of contamination in the environment where these animals are inserted. And it is concluded that the immune responses presented by the differences between the results of these parameters are indicative of changes in the water quality of the Neblina stream.
Keywords: Eastern Amazon, characid fish, blood, animal welfare.
INTRODUCTION
The lambari Astyanax novae Eigenmann, 1911, is a fish found in the eastern Amazon basin, with greater abundance in headwater rivers. It belongs to the Characidae family and the Tetragonopterinae subfamily and differs from the other congeneric species, except for the members of the A. bimaculatus group, a group of sympatric species from the same macro region. As A. novae is present in the São Francisco and Tocantins-Araguaia River drainages (FREITAS et al., 2015).
The infrequent geographic distribution of A. novae in the São Francisco and Tocantins-Araguaia River drainages is possible due to flooding episodes where the headwaters of the Sapão river (São Francisco River basin) and the headwaters of the Galhão river (Tocantins River drainage) forms a flooded complex of shared headlands. In this way, it is possible to exchange species in different hydrographic regions (FREITAS, 2011; LIMA and CAIRES, 2011; OLIVEIRA et al., 2017). This fact makes A. novae a widely distributed species, covering a large area of drainage and appearing in many environments and habitats (BARBOSA et al., 2019).
It is a relatively fast-growing and prolific fish like most representatives of the Characidae family. The species A. novae is considered omnivorous with insectivory tendencies, feeding on insects, small invertebrates and some fruits and seeds. It is a gregarious fish, forming from small groups to large schools, which prefer lotic and dystrophic environments, with oxygenated and clear water (FREITAS et al., 2011; 2015).
The study of hematological patterns is effective in determining pathological conditions in fish. However, the hematic composition also makes it possible to infer about the conditions of the environment where these organisms are inserted. Enabling the verification of environmental conditions, such as: temperature, salinity, oxygen, among others, as they interfere with the physiological conditions of hematological parameters (LIZAMA et al., 2020; STOSIK et al., 2020).
However, other factors also directly influence the conditions of hematological parameters, such as: malnutrition, age, size of this organism and seasonality; it may promote changes in the results of blood analyzes of these animals (TAVARES-DIAS et al., 2018; CICERO et al., 2020).
Analyzes of the variation of hematological parameters can be efficient tools for measuring and identifying specific environmental changes with rapid reflexes from environmental stress conditions (NOGUEIRA et al., 2020). However, these contributions to inferences about environmental contamination represent good effectiveness for short-term impacts (NASCIMENTO et al., 2020)
METHODOLOGY
DESCRIPTION OF THE STUDY AREA
This study was carried out in Neblina stream, an urban area in the municipality of Araguaína – TO, Brazil. Being located in the northern part of the state of Tocantins, it has a mixture of Brazilian ecosystems (Cerrado, Cocais Forest and Amazon Forest), forming a unique ecotone region (SAVIATO et al., 2020) (Figure 1, Figure 2 and Table 1). The region is marked by the environmental physiognomy, typical of the Cerrado biome, and the aforementioned river has riparian forest in much of its headwater (Figure 3), however the main channel is strongly inserted in the central region and suffers from urban pressures (Figure 4).
Table 1 – UTM geographic coordinates of the animal collection points within the Neblina stream catchment basin.
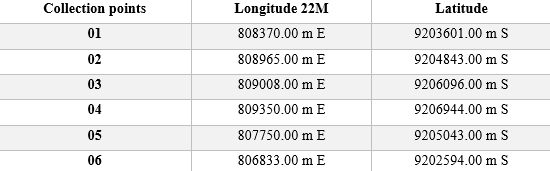
Figure 1 – Collection points inside located in the Neblina stream drainage basin, northern Tocantins.
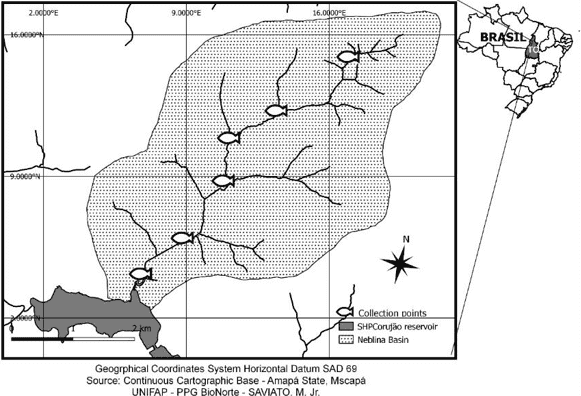
Figure 2 – Hydrographic network of the Neblina stream, strongly inserted in the urban center of Araguaína – TO, Brazil.
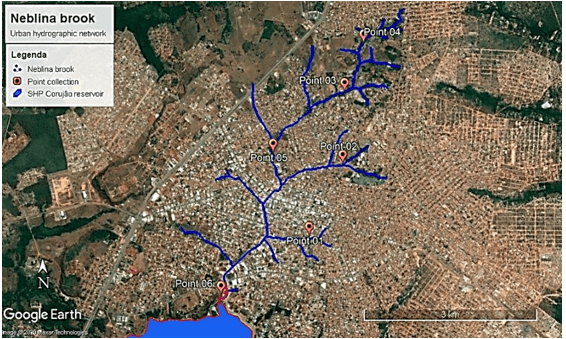
Figure 3 – Headland region of the Neblina stream upstream from the first collection point.

Figure 4 – Central region of the Neblina stream, undergoing a process of physiognomic change, for the construction of flood containment.

FISH COLLECTION
The catches of fish of the species A. novae (Figure 5), which is widely distributed in the headwaters of the Lontra River (SAVIATO et al., 2017; 2020) (Figure 5). were collected along the drainage basin of the Neblina stream, at the points listed in Table 1, in the central aria of the city of Araguaína – TO, Brazil. The collections take place in these 06 (six) points, distributed from the head of the aforementioned river to its mouth, which flows into the Lontra River where it is an important tributary (Figure 1, Figure 2 and Table 1). The study area exploration for this research took place in two seasons, dry and rainy, respectively, October 2020 and March 2021.
Figure 5 – Astyanax novae collected from the Neblina River in the Eastern Amazon.

These fish were collected with fishing methodologies, such as: cast net (5mm mesh between us and 2m radius); likewise, in places where cast nets were not possible, the Flyfishing methodology was invested, with a rod, reel and artificial bait (Figure 6). The collected animals then went through the blood extraction process and were subsequently anesthetized and euthanized with an Eugenol solution (50mg L-1).
Figure 6 – Animal captured with the methodology described with Flyfishing.

BLOOD COLLECTION
The fish of the species A. novae collected (red-tailed lambari) totaled 60 individuals with an average weight of 8.5 ± 0.3g. Immediately after collection, these animals had blood collected by puncture of the caudal vessels using a 1mL syringe, one containing 0.05ml of 10% EDTA, as an anticoagulant (Figure 7). Subsequently, hematological analyzes and blood smear slides were performed.
Figure 7 – Extraction of blood from A. novae by tail puncture.

BLOOD ANALYSIS METHODS
The hematological analyzes were performed at the Laboratory Núcleo de Saúde Bonamigo Ltda de Araguaína – TO (protocol nº 0219/2017 approved by the animal ethics committee) (Figure 8). During the procedures for hematological analyses, total hemocyte count were performed using the Neubauer hemocytometer and the hemoglobin concentration. Absolute hematimetric rates of mean corpuscular volume (MCV), mean corpuscular hemoglobin (MCH) and mean corpuscular hemoglobin concentration (MCHC) were also obtained. Blood smear slides were stained with Giemsa and May Grünwald (ROSENFELD, 1947), total and differential leukocyte counts were performed as described by Jatobá et al. (2011). And for immunological analysis Total Plasma Protein (Biochemical Reagent, Total Proteins, LabTest, Brazil); glucose (G-TECH free®, Accumed-Glicomed, Brazil) and total immunoglobulin concentration (SIWICKI and ANDERSON, 1993).
Figure 8 – Samples collected and in the process of laboratory analysis
ANALYSIS OF HEMATOLOGICAL DATA
The hematological data were subjected to Bartlett analysis, in order to ascertain the homogeneity of variance, ONE-WAY ANOVA, and the differences between treatments were then analyzed using the Student-Newman-Keuls (SNK) test. Thus, data on leukocyte, lymphocyte and monocyte counts were also submitted to regression analysis, as well as the coefficients assessed for significance were assessed by the t test. and finally all tests were carried out with a significance level of 5%.
RESULTS
HEMATOLOGICAL PARAMETERS
Blood analyzes in A. novae presented parameters with subtle discrepancies for each sampling site. These data were taken from 5 (five) animals per point per station, totaling the analysis of 10 animals for each sampling site (Table 2). Thus, from the compilation of data from all fish at all points, and analyzed by the statistical methods described, the results for each parameter were tabulated (Table 3).
It is noteworthy that the sequence of hematological parameters are quantitatively listed in Table 3: Leukocytes (GB)> Erythrocytes (GV)> Hemoglobin (Hb)> Hematocrit (Ht)> Mean Corpuscular Volume (MCV)> Mean Corpuscular Hemoglobin (HCM) > Hemoglobin Concentration (CHCM) > Erythrocyte Distribution Amplitude (ADVGVC) > Erythrocyte Distribution Amplitude Standard Deviation (ADVGSD) > Platelets (PLT) > Mean Platelet Volume (MPV) > Packet Distribution Amplitude (ADP) > Platetocrit (PCT).
Table 2 – Distribution of hematological parameter values and their means for each collection point in the Neblina stream, and the standard deviation (X±DE).
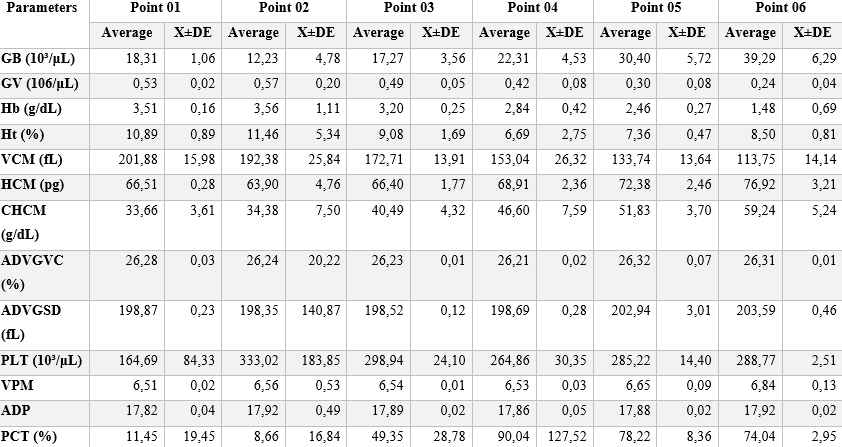
Table 3 – Hematological parameters and their means for Neblina stream, as a whole, and the standard deviation (X±DE) for each parameter analyzed.
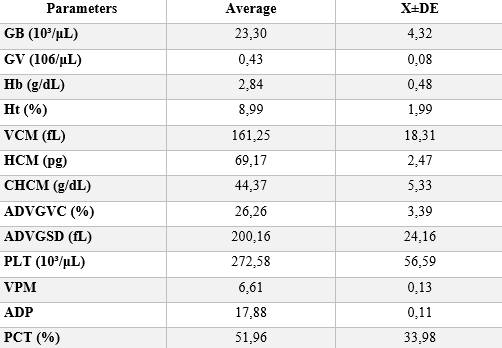
Figure 9 – Variation of hematological data for all samples, and their respective standard deviations.
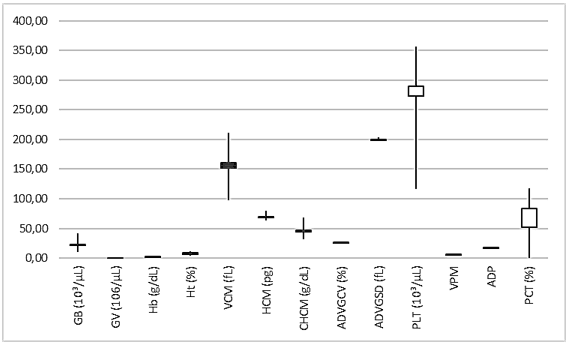
RED BLOOD CELL MORPHOLOGY
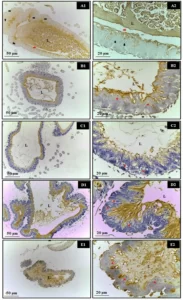
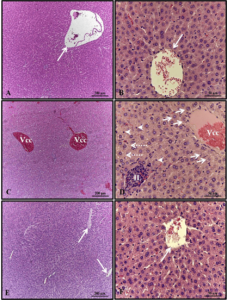
In the morphological evaluations of blood cells, it can be seen that the smear samples presented morphology and distribution of red and white cells in similar amounts and shape (Figure 9). However, in the samples from points 05 and 06, it was possible to identify some cells (on average 12.35% of the cells counted) with the presence of micronuclei. Presentation inherent to the presence of genotoxic agents in the environment (Figure 10).
Figure 10 – A. novae blood smear illustrating apparently healthy blood cells.
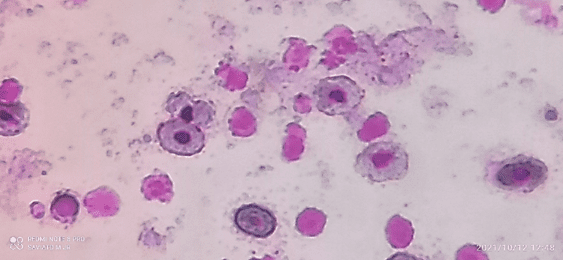
Figure 11 – A. novae blood smear, illustrating blood cells, with the presence of micronuclei (arrows).
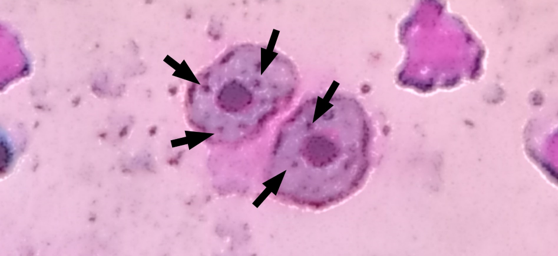
It is possible to observe that the red blood cells have a homogeneous general shape with few variations in the cytoplasm and shape of the nucleus. Leukocytes and Platelets have a variable shape, which is expected for fish. However, some leukocytes have a C-shaped nucleus, while most of the nucleus is ovoid and centralized in the cytoplasm. Noting that oval erythrocytes with transparent cytoplasm and very prominent, central and also oval nucleus.
DISCUSSION
In the experimental year, represented by two distinct climatic seasons, dry and rainy, hematological parameters were found that indicate significant differences between the hematological parameters for the described seasons. Among these parameters, it is possible to identify a greater relative variation in the concentration of Platelets (PCT = 51.96 ± 11.78%), while the smallest variation was the number of RBCs (GV = 0.43 ± 6.39 106/uL) (Table 3). Other parameters of importance in the analysis were the concentration indices for Hemoglobin (Hb = 3.63 ± 0.13 g/dL) and Total leukocytes (GB = 10.62 ± 3.63 103/uL), respectively indicating an increase in respiratory needs and the need to control infectious agents from the environment. This fact is corroborated by the encounter of urban solid waste and domestic effluent dumps, directly in the river. Affecting the fauna and flora as a whole, having evidence such as the colonization of the riverbed by cyanobacteria, having stretches with gray water and strong odor.
From the analysis of the slides and the results of the hematological parameters, it is possible to conclude that the data obtained are in line with the species of the genus Astyanax Baird & Girard, 1854, studied so far. With subtle deviations for these parameters in general and punctually in GB, GV, Hb and Ht, as described by Jatobá et al. (2020). As for Leukocytes, their count was consistent with that presented in the literature and indicated for other teleost fish (CASTRO et al., 2020; CICERO et al., 2020; STOSIK et al., 2020), except for some of the samples that showed numerical divergences with probable influence of water quality parameters (BARBOSA et al., 2019), which is visibly altered in the location as a result of urban evictions and environmental degradation (PEREIRA et al., 2019). For this study, it was not possible to distinguish the leukocytes from each other, only the general count, without distinguishing between Neutrophils, Lymphocytes, Monocytes, Eosinophils and Basophils, all of which were identified as Leukocytes. And a single group was considered, due to the difficulty of distinguishing between the Figuretive elements mentioned. However, in these analyses, the values found for VCM and HCM presented a significant difference between samples from different collection sites. The indices of GV, Hb, Ht and CHCM presented mean values lower than expected for the species (JATOBÁ et al., 2020).
The amount of EDTA in the syringes for sample removal can influence the dilution of the elements figured as a whole, which can influence the results obtained (DUMAN et al., 2019; NOGUEIRA et al., 2020).
It is possible that the procedures can exert a sensitive influence on the analyzes and their results. Even so, the results of this study are useful as a subsidy for other studies, even with different objectives, when using this same species for other environmental analyses.
CONCLUSION
Based on the analyzed data, we can identify that A. novae blood samples show differences in their respective hematological parameters. These differences are grouped and symmetrical for each sampling point. Indicating that there are pressures that lead such animals to undergo physiological changes reflected in hematological analyses. Considering the decrease in the hydrological volume presented for the dry season, it is possible that this factor corroborates the concentration of pollutants or toxic agents, differently for each sampling point. Thus, it can be said that these fish have different health conditions for each sampled environment. Being in homeostasis conditions closer to the headwaters, where the damage of urbanization or concentration of houses is also less visible, with the presence of better-preserved riparian forest.
FINAL CONSIDERATIONS
The species A. novae, due to its wide distribution in the studied basin, and in its hematological analyses, presents sensitive disparities visibly influenced by the environmental discrepancy between the collection points, it is possible that this species may reflect the environmental characteristics considered in the hematological parameters of the evaluated animals. The results presented in this work provide further studies and analyzes that imply decision-making to mitigate the environmental impacts resulting from urbanization. Facilitating the understanding of the effects of this urbanization on the natural environment, especially on the rivers that drain these areas and their fauna.
ACKNOWLEDGMENTS
To the Laboratory Núcleo de Saúde Bonamigo Ltda de Araguaína – TO, for the help with blood tests and availability of results; To the BioNorte Postgraduate Doctoral Program and its professors, for their support and encouragement; The authors are grateful to Rede BioNorte (Postgraduate Program in Biotechnology, Biodiversity and Conservation); and they also thank Forestry Engineer Lucca Pazini Moratelli, for his support and assistance in the preparation of thematic maps.
REFERENCES
BARBOSA HO, BORGES PP, DALA-CORTE RB, MARTINS PTA, & TERESA FB. 2019. Relative importance of local and landscape variables on fish assemblages in streams of Brazilian savanna. Fisheries Management and Ecology. 2019 (00): 1–12. https://doi.org/10.1111/fme.12331.
CASTRO PDS, LADISLAU DS, RIBEIRO MWS, LOPES ACC, LAVANDER HD, BASSUL LA, MATTOS DC, LIEBL ARS, ARIDE PHR & OLIVEIRA AT. 2020. Hematological parameters of three species of the peacock bass (Cichla spp.) from Balbina lake, Presidente Figueiredo, Amazonas, Brazil. Brazilian Journal of Biology, Ahead of Print, p01-07. https://doi.org/10.1590/1519-6984.219409
CICERO LH, SOUZA UP, ROTUNDO MM, PEREIRA CDS & SADAUSKAS-HENRIQUE H. 2020. Biometric and hematological indices of Mugil curema inhabiting two Neotropical estuaries. Regional Studies in Marine Science. 38(2020)101377: 01-08.
DUMAN IB, SATICIOGLU BS & ALTUN S. 2019. Drawing Blood Sample Practice from Teleost Fish. North American Journal of Aquaculture, 81(2): 119-125.
FREITAS MRM, LUCINDA PHF & LUCENA CAS. 2015. Redescription of Astyanax novae Eigenmann, 1911 (Teleostei: Characidae). Ichthyological Exploration of Freshwaters. 26(02): 183-192.
FREITAS MRM. 2011. Revisão taxonômica das espécies de Astyanax subgrupo goyacensis nas porções media e superior da drenagem do rio Tocantins. Unpublished MSc. dissertation, Universidade Federal do Tocantins, Porto Nacional. 47p.
JATOBÁ A, PEREIRA MO & RODHERMEL JCB. 2020. Hematological profile of Astyanax bimaculatus under different offer of Lactobacillus sp.. Arquivos Brasileiros de Medicina Veterinária e Zootecnia, 72(3): 871-878.
LIMA FCT & CAIRES RA. 2011. Peixes da Estação Ecológica Serra Geral do Tocantins, bacias dos rios Tocantins e São Francisco, com observações sobre as implicações biogeográficas das “águas emendadas” dos rios Sapão e Galheiros. Biota Neotropical, 11(1): 231-250.
LIZAMA M de los AP, CAGNI G dos S & ZAVASKI F. 2020. Análise histórica sobre a hematologia em peixes no Brasil: estudo quali/quantitativo. Centro Científico Conhecer – Jandaia-GO, 17(34):258-270.
NASCIMENTO, J. L. do, GIL, J. A., SMANIOTTO, F., VALLIM, J. H. & ISHIKAWA, M. M. 2020. Efeito das concentrações residuais de óleo cravo e do florfenicol nos biomarcadores hematológicos em tilápias. In: Congresso Interinstitucional de Iniciação Científica – CIIC 2020, 01/10 a 02/10 de 2020. Campinas – SP: Embrapa Informática Agropecuária. RE20403. 12p.
NOGUEIRA WV, OLIVEIRA FKde, GARCIA SdeO, SIBAJA KVM, TESSER MB & BUFFON JG. 2020. Fontes, técnicas de quantificação, riscos associados e medidas de controle de contaminação por micotoxinas da alimentação aquática. Critical Reviews in Microbiology – Taylor & Francis, p01-14. https://doi.org/10.1080/1040841X.2020.1716681
OLIVEIRA, CAM, PAVANELLI, CS, & BERTACO, VA. 2017. A new species of Astyanax Baird & Girard (Characiformes: Characidae) from the upper rio Araguaia, Central Brazil. Zootaxa, 4320 (1), 173. doi: 10.11646 / zootaxa.4320.1.10.
PEREIRA HR, GOMES LF, BARBOSA HdeO, PELICIE FM, NABOUT JC, TERESA FB & VIEIRA LCG. 2019. Research on dams and fishes: determinants, directions, and gaps in the world scientific production. SPRINGER – Hydrobiologia, 14p. https://doi.org/10.1007/s10750-019-04122-y
SAVIATO MJr, MARIANO WdosS, SAVIATO PLdoC & SASSI VB. 2017. Ictiofauna do ribeirão Jacubinha, bacia do rio Lontra na cidade de Araguaína-TO. ENCICLOPÉDIA BIOSFERA, Centro Científico Conhecer – Goiânia-GO, 14(25): 1362-1374.
SAVIATO MJr, MARIANO WdosS, SAVIATO PLdoC, SASSI VB, MARTINS PHO, PAULINO MG & ALMEIDA SSMdaSde. 2020. Fihes diversity in a reservoir of small hydroelectric power that suffers influence of urban evictions, in the city of Araguaína, Tocantins State, Brazil. FACIT Business and Technology Journal, 13(1):100-112.
STOSIK M, TOKARZ-DEPTULA B & DEPTULA W. 2020. Major histocompatibility complex in Osteichthyes. SCIENDO: Review Article. Journal Veterinary Research 64(2020) 127-136. Doi:10.2478/jvetres-2020-0025.
TAVARES MD, PAULINO MG, TEREZAN AP, FERNANDES JB, GIANI A & FERNANDES MN.. 2018. Biochemical and morphological biomarkers of the liver damage in the Neotropical fish, Piaractus mesopotamicus, injected with crude extract of cyanobacterium Radiocystis fernandoi. Environmental Science and Pollution Research. 25(16):15349–15356.
[1] Doctoral Student in Biotechnology and Biodiversity, Master in Environmental Engineering, Graduation in Biological Sciences. ORCID: https://orcid.org/0000-0002-8757-7915.
[2] Academic at the University Anhembi Morumbi, São Paulo, SP. ORCID: https://orcid.org/0000-0002-5516-5772.
[3] Student of Veterinary Medicine at the Faculty of Sciences of Tocantins – FACIT, Araguaína, TO. ORCID: https://orcid.org/0000-0002-5811-2625.
[4] Farmacêutico-Bioquímica, Pós-Graduação em Saúde Coletiva. ORCID: https://orcid.org/0000-0002-6558-9480.
[5] PhD in Biodiversity and Conservation Amazonas State University – Bionorte Network. ORCID: https://orcid.org/0000-0002-8233-2628.
[6] Project Manager of the State of Amapá Scientific and Technological Research Institute higher education institution to which the author is linked: IEPA-AP Unit: Macapá-AP.
Posted: October 2021.
Approved: November 2021.