ORIGINAL ARTICLE
DIOCESANO, Kamila Bezerra Fernandes [1], COSTA, Aurélio Antonio Ribeiro [2], LEITÃO, Glauber Moreira [3]
DIOCESANO, Kamila Bezerra Fernandes. COSTA, Aurélio Antonio Ribeiro. LEITÃO, Glauber Moreira. Multifocal/multicenter breast cancer: Clinical/epidemiological profile and immunohistochemical patterns among the foci: Cross-sectional study. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year 05, Ed. 06, Vol. 04, pp. 73-94. June 2020. ISSN: 2448-0959, Access link: https://www.nucleodoconhecimento.com.br/health/multifocal-multicentric, DOI: 10.32749/nucleodoconhecimento.com.br/health/multifocal-multicentric
SUMMARY
Multifocal/multicenter breast cancer (MF/MC) is a disease that comprises many paradigms in the current practice of breast cancer. To combat morbidity and mortality of this the best option is a rational therapeutic strategy, which should begin with a clear biological understanding of multicentricity and multifocality, since this is known, the correct treatment can be followed. Our objective was to define the profile of patients with multifocal and multicenter breast cancer and to evaluate whether there is disagreement between tumor foci regarding hormonal receptor and Her-2. We retrospectively analyzed 89 patients with multifocal and multicenter invasive breast cancer in two tertiary institutions in northeastern Brazil, evaluating hormonal receptor, Her-2 and ki67 in each tumor focus of 25 of these patients, highlighting disagreement or not between them, and type and histological grade, lymph node metastasis and distance and age. Of these patients, 9% presented heterogeneity between tumor foci. The proportion of metastasis in axillary lymph node was 42% and only 10% for distant metastasis. 45% of the women in the study had histological grade II or III. We found a prevalence of 40.5% of negative Her-2 and 45% of positive estrogen receptor. There was no statistically significant difference between multifocal and multicenter cancer. We conclude that MF/MC breast cancer shows heterogeneity between tumor foci in relation to biological parameters, which plays a crucial role in making decisions about treatment and consequently on tumor recurrence, prognosis and distant metastasis.
Keywords: Breast cancer, heterogeneity, multifocal, multicenter.
INTRODUCTION
Breast cancer is the malignant neoplasm that most affects women worldwide. It is the second leading cause of cancer death in developed countries, behind only lung cancer, and the leading cause of cancer death in developing countries. In 2013, there were about 1.8 million new cases worldwide and 471,000 deaths[1]. For Brazil, the estimate is approximately 60,000 new cases of breast cancer per year, with an estimated risk of 56.20 cases per 100,000 women. In the state of Pernambuco is 2,450 new cases, with a gross incidence rate of 90.25 cases per 100,000 inhabitants only for Recife, the capital[2].
Breast cancer is classified as multicenter (MC) and/or multifocal (MF) when it presents more than one tumoral, synchronous focus in the same breast. The difference between them is based on location, whether they are located in different quadrants of the breast or in it, respectivel[3,4]y.
The incidence of MF and/or MC tumors varies between 6% and 60[5,6]%. The clinical-pathological and prognostic characteristics of multifocal and multicenter breast cancer are not yet well established.
Based on the reasoning that lesions with similar histology and degree should be biologically similar, the American College of Pathology recommends characterizing all foci of MF/MC with immunohistochemistry only when histology and or histological degree differ from each other[7]. Therefore, immunohistochemistry is requested only for the larger tumor and the other foci are not evaluated[8].
A study including 113 patients, in which the immunohistochemistry of all tumor foci was evaluated, found that the disagreement between the foci was present in 4.4% – 18.6% of the cases, being indicated different treatments compared with what would have been prescribed if they had taken into account only the immunohistochemistry of a single focus, the larges[9]t.
The decision to choose the best therapeutic option for MC/MF breast cancer should begin with a clear biological and epidemiological understanding of multicentricity and multifocus, since these are known, the correct treatment can be followed. Immunohistochemical evaluation of tumors plays a crucial role in decision-making in the treatment of breast cancer and when expressed discordantly in the various foci may provide patients with the opportunity of different therapies.
According to what was studied, the objective of this study was to describe the clinical and epidemiological profile of women with MF/MC breast cancer and the immunohistochemical patterns of the various foci of breast cancer in each woman in two tertiary hospitals in northeastern Brazil.
METHODS
The descriptive cross-sectional study was carried out at the Instituto de Medicina Integral Prof. Fernando Figueira (IMIP) and the Barão de Lucena Hospital (HBL), institutions in the state of Pernambuco, focused on medical care in various specialties and committed to teaching, research and extension.
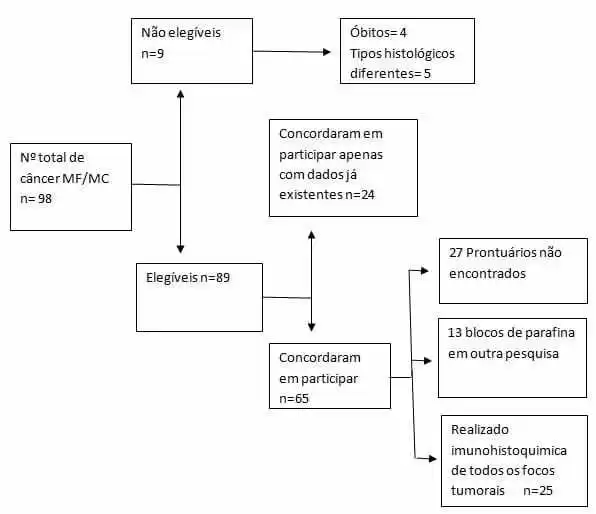
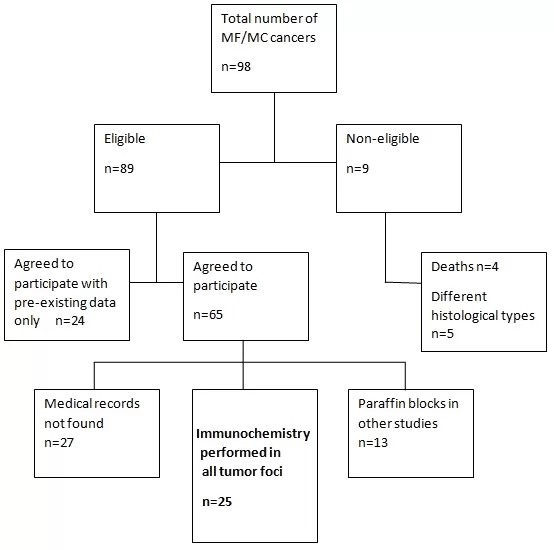
The sample was conventionally constituted by women retrospectively identified with MF or MC breast cancer assisted in the IMIP from January 2013 to June 2016 and in HBL in the period from January 2012 to June 2016 who meet the eligibility criteria of the research, which are: having two or more tumor foci diagnosed in the same breast simultaneously , except in cases of recurrence of a previous tumor, being alive and having the same type and histological degree between foci.
Weekly visits were made to the HBL pathological anatomy laboratory and the IMIP breast center and results of biopsy histopathological or surgical specimen of breast tumors of patients with a profile compatible with the eligibility criteria of the study were selected.
The tumor tissue obtained by core biopsy or at the time of definitive surgery is preserved and packed in formalin (10% formaldehyde) buffered and processed into paraffin blocks. These blocks are stored at room temperature until further analysis, both for histopathological and immunohistochemical examination.
After identifying these patients, the researcher contacted the patients who were presented to the research and invited to participate by signing the free and informed consent form.
Immunohistochemistry of each tumor focus was requested. This was obtained through the paraffin blocks already stored, or, retrospectively, from existing records of the tumor focus studied.
Immunohistochemistry is a vital tool in the study of breast lesions. Using specific antibodies against intracellular proteins present in a small amount in the nucleus, cytoplasm and/or cell membrane[10]. The immunohistochemical technique identifies in the tumor the estrogen and progesterone receptors, the tumor cell proliferation index and the expression of the HER-2 membrane protein. It was requested for all multiple tumors present simultaneously in the same breast and was performed in a laboratory with a professional specialized in breast pathology, in which it uses the method of streptavidin-biotin marked through the PT-link machine of DAKO.
Patients who did not authorize the request for immunohistochemistry agreed to participate in the study only with information contained in medical records and existing histopathological and immunohistochemical data.
Hormonal receptor, Her-2 and ki67 were evaluated in each tumor focus, highlighting disagreement or not between them; type and histological degree; lymph node metastasis and distance; lymphovascular invasion and age of the patient.
Figure 1. Participant capture flowchart
The information collected was entered in double entry (the researcher and a collaborator), at different times, in a data sheet (Microsoft Office Excel software – 2010), created with the purpose of validating typing. The database has undergone patch and cleanup testing to detect errors or inconsistencies in the data. Only after comparing the banks and performed the consistency and cleaning tests was the definitive database used for statistical analysis.
The Software SPSS 13.0 (Statistical Package for the Social Sciences) for Windows and Excel 2010 were used for data analysis. To analyze the behavior of the study variables, measures of central tendency and their dispersions were performed, the Quiquadrado test of trends and Fisher’s Exact, at a significance level of 5%, when relevant. All tests were applied with 95% confidence. The results are presented in table form with their respective absolute and relative frequencies.
RESULTS
The incidence of MF/MC tumors in our study was 4% in 2013 and 5% in 2015. Imip data for 2012 and 2014 are not available (Table 1). 64% of women with MF/MC cancer were over 50 years of age. With the exception that many data were not available, we found a frequency of 33% of brown women, 24% with incomplete elementary school and 13% of married women (Table 2).
TABLE 1 – Incidence of unifocal, multifocal and multicenter breast cancer at Hospital Barão de Lucena, HBL, and Instituto Materno Infantil Prof. Fernando Figueira, IMIP. Recife, Pernambuco
| CA de Mama | n (HBL/IMIP) | % (HBL/IMIP) |
| 2012 | ||
| Unifocal, California | (195/-) | (92,4/-) |
| MC/MF | (16/-) | (7,6/-) |
| 2013 | ||
| Unifocal, California | 477 (184/293) | 95,9 (93,4/97,7) |
| MC/MF | 20 (13/ 7) | 4,0 (6,6/2,3) |
| 2014 | ||
| Unifocal, California | (150/-) | (92,6/-) |
| MC/MF | (12/12) | (7,4/-) |
| 2015 | ||
| Unifocal, California | 523 (162/361) | 94,9 (89,5/97,6) |
| MC/MF | 28 (19/9) | 5,0 (10,5/2,4) |
IMIP data for 2012 and 2014 are not available.
Source: IMIP/HBL Search
TABLE 2 – Characteristics of patients with multifocal and multicenter breast cancer operated at Hospital Barão de Lucena, HBL, and Instituto Materno Infantil Prof. Fernando Figueira, IMIP. Recife, Pernambuco
| Variables | n | % |
| Age | ||
| < 50 | 26 | 29,2 |
| ≥ 50 | 57 | 64,1 |
| Cor | ||
| Branca | 16 | 18,0 |
| Black | 2 | 2,2 |
| Brown | 30 | 33,7 |
| Schooling | ||
| Illiterate | 6 | 6,7 |
| Incomplete elementary school | 22 | 24,7 |
| Complete elementary school | 6 | 6,7 |
| Incomplete high school | 1 | 1,1 |
| Complete high school | 12 | 13,5 |
| Marital Status | ||
| Stable union | 1 | 1,1 |
| Single | 8 | 9,0 |
| Married | 13 | 14,6 |
| Divorced | 3 | 3,4 |
| Widow | 9 | 10,1 |
The uninformed data for each variable were omitted from the table.
Source: IMIP/HBL Search
Ductal histological type was found in 82% of women in our study. The proportion of metastasis in axillary lymph node was 42% and only 10% for distant metastasis. 45% of the women in the study had histological grade II or III. We found a prevalence of 40.5% of negative Her-2 and 45% of positive estrogen receptor. As for Ki67, we found 41% with a high rate of cell proliferation. The proportion of heterogeneity between foci was 9% and we found 65% of MF and 17% of MC (Table 3).
TABLE 3 – Clinical and tumor characteristics of patients with multifocal and multicenter breast cancer operated at Barão de Lucena Hospital, HBL, and Instituto Materno Infantil Prof. Fernando Figueira, IMIP. Recife, Pernambuco
| Variables | n | % |
| Re | ||
| Positive | 40 | 44,9 |
| Negative | 20 | 22,5 |
| RP | ||
| Positive | 35 | 39,3 |
| Negative | 25 | 28,1 |
| Her-2 | ||
| Positive | 18 | 20,2 |
| Negative | 36 | 40,5 |
| Ki67 | ||
| <14% | 16 | 18,0 |
| >14% | 37 | 41,6 |
| Compromised lymph nodes | ||
| Out | 23 | 25,8 |
| 1 a 3 | 18 | 20,2 |
| 4 or more | 20 | 22,5 |
| Histological degree | ||
| I | 9 | 10,1 |
| II | 27 | 30,4 |
| III | 13 | 14,6 |
| Histological type | ||
| Ductal | 73 | 82,0 |
| Lobular | 1 | 1,1 |
| Other | 4 | 4,5 |
| Size of the largest tumor | ||
| <2 cm | 24 | 27,0 |
| 2-5 cm | 32 | 36,0 |
| >5 cm | 5 | 5,6 |
| Distant metastasis | ||
| Yes | 9 | 10,1 |
| No | 48 | 53,9 |
| Lymphovascular invasion | ||
| Yes | 20 | 22,5 |
| No | 24 | 27,0 |
| Heterogeneity between foci | ||
| Yes | 8 | 9,0 |
| No | 17 | 19,1 |
| Distance between foci | ||
| ≤ 4 cm (multifocal) | 58 | 65,2 |
| > 4 cm (multicenter) | 15 | 16,9 |
The uninformed data for each variable were omitted from the table.
Source: IMIP/HBL Search
No significant differences were observed in the comparison of variables between women with or without heterogeneity between tumor foci (Table 4) and between MF and MC (Table 5).
TABLE 4 – Clinical and tumor characteristics of patients with multifocal/multicenter breast cancer operated at Barão de Lucena Hospital, HBL, and Instituto Materno Infantil Prof. Fernando Figueira, IMIP, in relation to the heterogeneity between foci. Recife, Pernambuco
| Heterogeneity between foci | |||
| Variables | Yes | No | p-value |
| n (%) | n (%) | ||
| Compromised lymph nodes | |||
| Out | 2 (40,0) | 3 (60,0) | 1,000 * |
| 1 a 3 | 2 (33,3) | 4 (66,7) | |
| 4 or more | 3 (50,0) | 3 (50,0) | |
| Histological degree | |||
| I | 2 (66,7) | 1 (33,3) | 0,748 * |
| II | 2 (25,0) | 6 (75,0) | |
| III | 1 (33,3) | 2 (66,7) | |
| Size of the largest tumor | |||
| <2 cm | 4 (57,1) | 3 (42,9) | 0,510 * |
| 2-5 cm | 3 (33,3) | 6 (66,7) | |
| >5 cm | 0 (0,0) | 2 (100,0) | |
| Distant metastasis | |||
| Yes | 1 (20,0) | 4 (80,0) | 0,588 * |
| No | 5 (45,5) | 6 (54,5) | |
| Lymphovascular invasion | |||
| Yes | 4 (57,1) | 3 (42,9) | 0,592 * |
| No | 2 (28,6) | 5 (71,4) | |
(*) Fisher’s Exact Test
Source: IMIP/HBL Search
TABLE 5 – Clinical and tumor characteristics of patients with multifocal and multicenter breast cancer operated at Barão de Lucena Hospital, HBL, and Instituto Materno Infantil Prof. Fernando Figueira, IMIP. Recife, Pernambuco
| Distance between foci | |||
| Variables | ≤4cm (multifocal) | >4cm (multicenter) | p-value |
| n (%) | n (%) | ||
| Re | |||
| Positive | 29 (80,6) | 7 (19,4) | 1,000 * |
| Negative | 12 (80,0) | 3 (20,0) | |
| Rp | |||
| Positive | 26 (81,3) | 6 (18,7) | 1,000 * |
| Negative | 15 (78,9) | 4 (21,1) | |
| Her-2 | |||
| Positive | 14 (82,4) | 3 (17,6) | 0,723 * |
| Negative | 22 (75,9) | 7 (24,1) | |
| Ki67 | |||
| <14% | 13 (86,7) | 2 (13,3) | 0,695 * |
| >14% | 22 (75,9) | 7 (24,1) | |
| Compromised lymph nodes | |||
| Out | 16 (84,2) | 3 (15,8) | 0,839 * |
| 1 to 3 | 11 (73,3) | 4 (26,7) | |
| 4 or more | 16 (80,0) | 4 (20,0) | |
| Histological degree | |||
| I | 6 (66,7) | 3 (33,3) | 0,440 * |
| Ii | 20 (87,0) | 3 (13,0) | |
| Iii | 9 (81,8) | 2 (18,2) | |
| Histological type | |||
| Ductal | 49 (80,3) | 12 (19,7) | 0,370 * |
| Lobular | 1 (100,0) | 0 (0,0) | |
| Other | 2 (50,0) | 2 (50,0) | |
| Size of the largest tumor | |||
| <2 cm (1.5 in) | 19 (79,2) | 5 (20,8) | 0,884 * |
| 2-5 cm | 20 (76,9) | 6 (23,1) | |
| >5 cm | 2 (66,7) | 1 (33,3) | |
| Distant metastasis | |||
| Yes | 5 (62,5) | 3 (37,5) | 0,331 * |
| No | 35 (83,3) | 7 (16,7) | |
| Lymphovascular invasion | |||
| Yes | 13 (72,2) | 5 (27,8) | 0,256 * |
| No | 21 (87,5) | 3 (12,5) | |
(*) Fisher Exact Test Source: IMIP/HBL Research
DISCUSSION
The incidence of MF/MC tumors was approximately 5%, being 65% MF and 17% MC, slightly below that found in the litera[5,6,9]ture, which most likely stems from the fact that this rate varies according to the extent of breast tissue sampling analyzed by the pathologist on macroscopy, since it was demonstrated that a more complete sampling resulted in the identification of additional lesions; with the lack of a standard MF/MC [5]definition; and with the different sensitivities of imaging methods used preoperatively to detect multiple malignant foci[3,4,11-13]. An American study[5] evaluated 3,924 women with breast cancer and found 24% of MF/MC using only pathological criteria in their selection, 79% MF and 20% MC. A meta-analysis including 22 studies and 67557 women found MF/MC in 9.5% of cases, but some studies used preoperative imaging methods as a diagnostic criterion[14].
We found a larger sample of brown/black and married women. The low school level observed in this study reflects the patient’s degree of knowledge, which increases as schooling increases. A Brazilian study sh[15]owed an association between knowledge about risk factors for breast cancer and years of study, and there was no relationship between the variables skin color and marital status. An American study a[16]ssociated Afro-descendant women and never having been married, with advanced stage of the disease, perhaps because these women lacked the financial and social support provided by the spouse and by cultural and socioeconomic factors related to color. In our study, although we had a prevalence of women with low schooling, brown and black, we did not observe an increase in advanced stage.
Most patients in the study were 50 years or older (64%). It is known that breast cancer is more frequent in this age group, and this is a possible explanation for this sample. Some reports in the literature associate MF/MC with young women under 50 years of age, lob[3,12]ular histological type an[17]d greater lymph node involvement[3,4,8,12,17], supporting the hypothesis that MF/MC are biologically more aggressive, have a greater propensity to metastatic diffusion and are related to a worse result[12].
The proportion of axillary lymph nodes affected in our study was 42% and only 10% for distant metastasis. Patients with multiple invasive carcinomas are at higher risk of metastasis in the axillary lymph nodes, however, it has not been shown to increase the risk of distant metastasis for patients with negative ganglion disease[4]. An Australian study wi[18]th 848 women with unifocal breast cancer and MF/MC performed multivariate analysis comparing several variables with the measurement of the size of the largest tumor focus and with the measurement of the sum of the various tumor foci, and concluded that the tendency of metastasis of breast tumors is a reflection of the total tumor load instead of the dimension of the greatest focus and that the use of the cumulative size of foci in multifocal tumors demonstrates a lymph node positivity identical to that of the equivalent size of unifocal tumors. Using the size of the dominant focus, there is a higher probability of lymph node positivity and perhaps a worse prognosis compared to unifocal tumors of the same size.
The most frequent histological type was ductal (82%), similar to that found by several authors[3,9,17,19]. Lynch et al[5] describe an increase in the rate of metastases to regional lymph nodes in relation to unifocal tumors. This association raises the question of whether MF/MC are underestimated with the current staging system, which takes into account only the larger tumor focus, or if they simply have a more aggressive inherent biology.
Histological degree is a known prognostic factor in breast cancer. Numerous studies have demonstrated its significant association with survival. At the same time, it is an important component of therapeutic decision and has a predictive role in therapeutic respons[20]e. Studies show a strong association between MF/MC and histological grade II/II[11,17,19]I. 45% of the women in the study had histological grade II or III.
We found a prevalence of 40.5% of negative Her-2 and 45% of positive estrogen receptor, against data in the literature that show a correlation between MF/MC and pathological clinical factors suggestive of more aggressive tumors, with remarkable association of positive Her2 and absence of hormonal estrogen receptor[12]. We believe that the cause of this difference may be due to the small sample size, thus characterizing a type II statistical error. Regarding Ki67, 41% corresponded to a high cell proliferation. Indeed, some cases of hormone-responsive tumors will be conducted to chemotherapy due to this fact.
The American College of Pathology[7] considers it sufficient that the immunohistochemical characterization of multifocal lesions with similar degree and histology is based only on the largest lesion. However, recent studies que[9,21,22]stion this recommendation, as they have demonstrated that, despite the similarity of pathological characteristics, the lesions may be different in terms of estrogen receptor status and Her-2, potentially affecting their treatment. In our study, we found 9% of heterogeneity between foci, including ki67 analysis. Buggi et [9]al found discrepancy between foci in relation to Ki67 in 15% of cases.
Hormonal receptors were discrepant between foci in 8% of the sample and Her-2 in 4%, but the analysis of these items was impaired due to the fact that in more than 50% of the cases immunohistochemistry was obtained only from the highest tumor focus. Buggi et [9]al described a disagreement rate between hormone receptor results for estrogen of 4.4% and Her-2 of 9.7% among foci. Peka[22]r and C[21]hoi observed discrepancy rates between 3-4% for estrogen receptor and 6% for Her-2. According to[22] Pekar, patients who demonstrated heterogeneity had a worse prognosis compared to patients who did not demonstrate heterogeneity, which was not found in our study, in which no significant differences were observed between the heterogeneity and homogeneous groups, but we had limitations due to the small sample and losses in the analysis, which could change this scenario.
There was no statistical significance in the comparison of variables between MF and MC due to a small sample size. In the literature, we did not observe large differences between MF/MC in relation to type and histological degree, lymph node metastasis and tumor distance and siz[5,13]e.
Clinical decisions in systemic adjuvant therapy in breast cancer are based on histological criteria and immunohistochemical profile of the greater tumor focus, ignoring the smaller simultaneous foci.
The presence of additional foci is not taken into account in the decision of adjuvant therapies. If we examine only the larger tumor in such cases, we may lose patients who needed specific therapy. The consequences of heterogeneity between tumor foci for therapeutic decision-making and, therefore, for increased patient survival are rarely addressed in the literature.
Therefore, greater attention should be paid to MF/MC tumors and the possibility of having as routine the request for immunohistochemistry for all tumor foci should be routinely evaluated, since by the standard approach patients with disagreement between foci would have stopped receiving hormonal and anti Her-2 treatment for breast cancer.
There are some limitations to our analysis. First, our work was retrospective, although some of the approaches of the extracted data were requested during the study, data on histopathological tumor could not be detailed, when they were not made at the time of the preparation of the histopathological result; in addition to the fact that some paraffin blocks were available for further research, making it impossible to perform immunohistochemistry and, consequently, a small sample size. This fact may fail to highlight some associations that might exist, thus characterizing a type II statistical error. This fact impairs the external validation of our results.
CONCLUSION
Most MF/MC cancers have homogeneity among tumor foci, and evaluating more than one focus can have therapeutic consequences in only a minority of cases, in addition to increasing cost and time. However, since the prognosis and treatment of breast cancer patients may vary according to the molecular characterization of the tumor, it can be argued, from the biological point of view, that analyzing only one focus, as indicated in the current recommendations, would not be appropriate. There is emerging evidence that this recommendation may not be sustainable over time and may need to be adapted to be compatible with the evolution of breast cancer paradigms.
We conclude that MF/MC breast cancer shows a small proportion of heterogeneity between foci in relation to biological parameters, however, this plays a crucial role in making decisions about adjuvant treatment and consequently about the rate of prognosis, tumor recurrence and distant metastasis for women with such finding. It would be extremely interesting to routinely request immunohistochemistry for all tumor foci in cases of MF/MC. However, considering the potential additional financial burden for laboratories when instituting this recommendation, pathology laboratories may need to develop additional strategies to deal with this new proposal.
ETHICAL ASPECTS
This project complies with the current laws of the country, and the postulates of the Declaration of Helsinki,[23] being approved by the local Ethics and Research Committee (CEP).
REFERENCES
- Global Burden of Disease Cancer Collaboration. “The Global Burden of Cancer 2013.” JAMA oncology4 (2015): 505–527.
- MS/INCA. Estimativa 2016: Incidência de Câncer no Brasil/ Instituto Nacional de Câncer José Alencar Gomes da Silva, Coordenação de prevenção e vigilância. Rio de Janeiro: INCA, 2015. ISBN 978-85-7318-283-5, acessado em inca.gov.br/estimativa/2016/estimativa-2016-v11.pdf
- Zhou M, Tang Z, Li J, Fan J H, Pang Y, et al; Clinical and Pathologic Features of Multifocal and Multicentric Breast Cancer in Chinese Women: A Retrospective Cohort Study. Journal of Breast Cancer. 2013;16(1):77-83.
- Salgado R, Aftimos P, Sotiriou C, Desmedt C; Evolving paradigms in multifocal breast cancer. Semin Cancer Biol. 2015 Apr; 31:111-8.
- Lynch S P, Lei X, Chavez-MacGregor M, Hsu L, Meric-Bernstam F, et al; Multifocality and multicentricity in breast cancer and survival outcomes. Annals of Oncology 2012, 23: 3063–3069
- Lynch S P, Lei X, Hsu L, Meric-Bernstam F, Buchholz T A, et al; Breast Cancer Multifocality and Multicentricity and Locoregional Recurrence. The Oncologist 2013, 18:1167-1173.
- Fitzgibbons P L, Bose S, Chen Y, Connolly J L, Baca M E et al. Protocol for the examination of specimens from patients with invasive carcinoma of the breast. Version Invasivebreast 4.0.0.0 June 2017 cap.org/cancerprotocols 2017 College of American Pathologists (CAP)
- Weissenbacher T M, Zschage M, Janni W, Jeschke U, Dimpfl T, et al; Multicentric and multifocal versus unifocal breast cancer: is the tumor-node-metastasis classification justified? Breast Cancer Res Treat (2010) 122:27–34
- Buggi F, Folli S, Curcio A, Casadei-Giunchi D, Rocca A, et al; Multicentric/multifocal breast cancer with a single histotype: is the biological characterization of all individual foci justified? Annals of Oncology 2012 23: 2042–2046
- Chagas C R, et al. Tratado de Mastologia da SBM – Rio de Janeiro : Revinter, 2011. ISBN 978853720346-0
- Boros M, Podoleanu C, Georgescu R, Moldovan C, Molna C, et al; Multifocal/multicentric breast carcinomas showing intertumoral heterogeneity: a comparison of histological tumor and lymph node metastasis. Pol J Pathol 2015; (2): 125-132.
- Neri A, Marrelli D, Megha T, Bettarini F, Tacchini D, et al. “Clinical significance of multifocal and multicentric breast cancers and choice of surgical treatment: a retrospective study on a series of 1158 cases.” BMC Surgery. 2015;15(1):1.
- Kadioğlu H, Özbaş S, Akcan A, Soyder A, Soylu L, et al. Comparison of the histopathology and prognosis of bilateral versus unilateral multifocal multicentric breast cancers. World Journal of Surgical Oncology. 2014; 12:266.
- Vera-Badillo F E, Napoleone M, Ocana A, Templeton A J, Seruga B, et al.; Effect of multifocality and multicentricity on outcome in early stage breast cancer: a systematic review and meta-analysis. BreastCancer Res Treat 2014; 146: 235-244.
- Batiston A P, Tamaki E M, Souza L A, Santos M L M. ; Conhecimento e prática sobre os fatores de risco para o câncer de mama entre mulheres de 40 a 69 anos. RevBras Saúde MaternInfant, Recife; 2011; 11 (2): 163-171.
- Lannin D R, Mathews H F, Mitchell J; Influence of socioeconomic and cultural factors on racial differences in late stage presentation of breast cancer. JAMA. 1998; 279 (22): 1801-1807.
- Yerushalmi R, Kennecke H, Woods R, Olivotto I A, Speers C, et al. Does multicentric/multifocal breast cancer differ from unifocal breast cancer? An analysis of survival and contralateral breast cancer incidence. Breast Cancer Res Treat (2009) 117: 365.
- Coombs J, Boyages J; Multifocal and multicentric breast cancer: does each focus matter? J ClinOncol 2005; 23: 7497-7502.
- M, Marian. C, Moldovan. C, Stolnicu. S, Morphological heterogeneity of the simultaneous ipsilateral invasive tumor foci in breast carcinoma: A retrospective study of 418 cases of carcinomas, In Pathology – Research and Practice, 2012, (208): 604-609, ISSN 0344-0338.
- Curigliano G, Burstein H J, Winer E, Gnant M, Dubsky P, et al. De-escalating and escalating treatments for early-stage breast cancer: the St. Gallen International Expert Consensus Conference on the Primary Therapy of Early Breast Cancer 2017. Ann Oncol.2017 Aug 1;28(8):1700-1712.
- Choi Y, Kim E J, Seol H, Lee H E, Jang M J, et al. The hormone receptor, human epidermal growth factor receptor 2, and molecular subtype status of individual tumor foci in multifocal/multicentric invasive ductal carcinoma of breast. Hum Pathol.2012 Jan;43(1):48-55.
- Pekar, G., Gere, M., Tarjan, M., Hellberg, D. and Tot, T., Molecular phenotype of the foci in multifocal invasive breast carcinomas: Intertumoral heterogeneity is related to shorter survival and may influence the choice of therapy. Cancer, 2014 Jan 1; 120 (1): 26–34.
- Review C, Communication S, Principles G. World Medical Association Declaration of Helsinki: ethical principles for medical research involving human subjects. JAMA. 2013;310:2191-2194.
APPENDIX – FIGURES AND TABLES IN ENGLISH
Fig. 1 Flow chart of participant selection
TABLE 1 – Incidence of unifocal, multifocal, and multicentric breast cancer at the Baron de Lucena Hospital (HBL) and Prof. Fernando Figueira Mother and Child Institute (IMIP). Recife, Pernambuco
| Breast cancer | n (HBL/IMIP) | % (HBL/IMIP) |
| 2012 | ||
| Unifocal | (195/-) | (92.4/-) |
| MC/MF | (16/-) | (7.6/-) |
| 2013 | ||
| Unifocal | 477 (184/293) | 95.9 (93.4/ 97.7) |
| MC/MF | 20 (13/7) | 4.0 (6.6/ 2.3) |
| 2014 | ||
| Unifocal | (150/-) | (92.6/-) |
| MC/MF | (12/ 12) | (7.4/-) |
| 2015 | ||
| Unifocal | 523 (162/361) | 94.9 (89.5/ 97.6) |
| MC/MF | 28 (19/ 9) | 5.0 (10.5/ 2.4) |
IMIP data for 2012 and 2014 are not available. Source: IMIP/HBL research
TABLE 2 – Characteristics of patients with multifocal and multicentric breast cancer operated at the Barão de Lucena Hospital (HBL) and Prof. Fernando Figueira Mother and Child Institute (IMIP). Recife, Pernambuco
| Variables | n | % |
| Age | ||
| <50 | 26 | 29.2 |
| ≥50 | 57 | 64.1 |
| Skin color | ||
| White | 16 | 18.0 |
| Black | 2 | 2.2 |
| Mixed race black | 30 | 33.7 |
| Schooling | ||
| Illiterate | 6 | 6.7 |
| Incomplete elementary school | 22 | 24.7 |
| Complete elementary school | 6 | 6.7 |
| Incomplete high school | 1 | 1.1 |
| Complete high school | 12 | 13.5 |
| Marital status | ||
| Civil union | 1 | 1.1 |
| Single | 8 | 9.0 |
| Married, New Year | 13 | 14.6 |
| Divorced | 3 | 3.4 |
| Widowed | 9 | 10.1 |
Data not reported for each variable were omitted from the table. Source: IMIP/HBL research
TABLE 3 – Clinical and tumor characteristics of patients with multifocal and multicentric breast cancer operated at the Barão de Lucena Hospital (HBL) and Prof. Fernando Figueira Mother and Child Institute (IMIP). Recife, Pernambuco
| Variables | n | % |
| ER | ||
| Positive | 40 | 44.9 |
| Negative | 20 | 22.5 |
| PR | ||
| Positive | 35 | 39.3 |
| Negative | 25 | 28.1 |
| HER2 | ||
| Positive | 18 | 20.2 |
| Negative | 36 | 40.5 |
| Ki67 | ||
| <14% | 16 | 18.0 |
| >14% | 37 | 41.6 |
| Compromised lymph nodes | ||
| Absent | 23 | 25.8 |
| 1 to 3 | 18 | 20.2 |
| 4 or more | 20 | 22.5 |
| Histological grade | ||
| I | 9 | 10.1 |
| II | 27 | 30.4 |
| III | 13 | 14.6 |
| Histological type | ||
| Ductal | 73 | 82.0 |
| Lobular | 1 | 1.1 |
| Others | 4 | 4.5 |
| Size of the largest tumor | ||
| <2 cm | 24 | 27.0 |
| 2-5 cm | 32 | 36.0 |
| >5 cm | 5 | 5.6 |
| Distant metastasis | ||
| Yes | 9 | 10.1 |
| No | 48 | 53.9 |
| Lymphovascular invasion | ||
| Yes | 20 | 22.5 |
| No | 24 | 27.0 |
| Heterogeneity between foci | ||
| Yes | 8 | 9.0 |
| No | 17 | 19.1 |
| Distance between foci | ||
| ≤ 4 cm (multifocal) | 58 | 65.2 |
| > 4 cm (multicentric) | 15 | 16.9 |
Data not reported for each variable were omitted from the table. ER: Estrogen receptor PR: Progesterone receptor. Source: IMIP/HBL research
TABLE 4 – Clinical and tumor characteristics of patients with multifocal/multicentric breast cancer operated at the Barão de Lucena Hospital (HBL) and Prof. Fernando Figueira Mother and Child Institute (IMIP), in relation to heterogeneity between foci. Recife, Pernambuco
| Heterogeneity between foci | |||
| Variables | Yes | No | p-value |
| n (%) | n (%) | ||
| Compromised lymph nodes | |||
| Absent | 2 (40.0) | 3 (60.0) | 1.000 * |
| 1 to 3 | 2 (33.3) | 4 (66.7) | |
| 4 or more | 3 (50.0) | 3 (50.0) | |
| Histological grade | |||
| I | 2 (66.7) | 1 (33.3) | 0.748 * |
| II | 2 (25.0) | 6 (75.0) | |
| III | 1 (33.3) | 2 (66.7) | |
| Size of the largest tumor | |||
| <2 cm | 4 (57.1) | 3 (42.9) | 0.510 * |
| 2-5 cm | 3 (33.3) | 6 (66.7) | |
| >5 cm | 0 (0.0) | 2 (100.0) | |
| Distant metastasis | |||
| Yes | 1 (20.0) | 4 (80.0) | 0.588 * |
| No | 5 (45.5) | 6 (54.5) | |
| Lymphovascular invasion | |||
| Yes | 4 (57.1) | 3 (42.9) | 0.592 * |
| No | 2 (28.6) | 5 (71.4) | |
(*) Fisher’s Exact Test. Source: IMIP/HBL research
TABLE 5 – Clinical and tumor characteristics of patients with multifocal and multicentric breast cancer operated at the Barão de Lucena Hospital (HBL) and Prof. Fernando Figueira Mother and Child Institute (IMIP). Recife, Pernambuco
| Distance between foci | |||
| Variables | ≤4 cm (multifocal) | >4 cm (multicentric) | p-value |
| n (%) | n (%) | ||
| ER | |||
| Positive | 29 (80.6) | 7 (19.4) | 1.000 * |
| Negative | 12 (80.0) | 3 (20.0) | |
| PR | |||
| Positive | 26 (81.3) | 6 (18.7) | 1.000 * |
| Negative | 15 (78.9) | 4 (21.1) | |
| HER2 | |||
| Positive | 14 (82.4) | 3 (17.6) | 0.723 * |
| Negative | 22 (75.9) | 7 (24.1) | |
| Ki67 | |||
| <14% | 13 (86.7) | 2 (13.3) | 0.695 * |
| >14% | 22 (75.9) | 7 (24.1) | |
| Compromised lymph nodes | |||
| Absent | 16 (84.2) | 3 (15.8) | 0.839 * |
| 1 to 3 | 11 (73.3) | 4 (26.7) | |
| 4 or more | 16 (80.0) | 4 (20.0) | |
| Histological grade | |||
| I | 6 (66.7) | 3 (33.3) | 0.440 * |
| II | 20 (87.0) | 3 (13.0) | |
| III | 9 (81.8) | 2 (18.2) | |
| Histological type | |||
| Ductal | 49 (80.3) | 12 (19.7) | 0.370 * |
| Lobular | 1 (100.0) | 0 (0.0) | |
| Others | 2 (50.0) | 2 (50.0) | |
| Size of the largest tumor | |||
| <2 cm | 19 (79.2) | 5 (20.8) | 0.884 * |
| 2-5 cm | 20 (76.9) | 6 (23.1) | |
| >5 cm | 2 (66.7) | 1 (33.3) | |
| Distant metastasis | |||
| Yes | 5 (62.5) | 3 (37.5) | 0.331 * |
| No | 35 (83.3) | 7 (16.7) | |
| Lymphovascular invasion | |||
| Yes | 13 (72.2) | 5 (27.8) | 0.256 * |
| No | 21 (87.5) | 3 (12.5) | |
(*) Fisher’s Exact Test. Source: IMIP/HBL research
[1] Master in maternal and child health by the stricto sensu graduate program of the Instituto de Medicina Integral Professor Fernando Figueira – IMIP, Pernambuco.
[2] PhD in Tocoginecology from the State University of Campinas, Brazil.
[3] Master’s degree in Oncology from the University of São Paulo, Brazil.
Sent: February, 2020.
Approved: June, 2020.