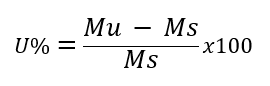ORIGINAL ARTICLE
ALMEIDA, Hellen Karine Santos [1], PAIVA, Matheus Monteiro Ybanez [2], DIAS, Claudio Alberto Gellis de Mattos [3], FECURY, Amanda Alves [4], DENDASCK, Carla Viana [5], DANTAS, Antônio de Pádua Arlindo [6]
ALMEIDA, Hellen Karine Santos. Et al. Technological analysis of material from Areal Morro Branco, Porto Grande, Amapá, by fractionation. Revista Científica Multidisciplinar Núcleo do Conhecimento. Year 05, Ed. 03, Vol. 11, pp. 05-13. March 2020. ISSN: 2448-0959, Access Link: https://www.nucleodoconhecimento.com.br/environment/technological-analysis, DOI: 10.32749/nucleodoconhecimento.com.br/environment/technological-analysis
SUMMARY
Natural sand is extracted using mining methods, even for the manufacture of artificial sands. Sand is widely used in several areas. As construction aggregates; material processing industries; water and sewage treatment. The Areal of the company Morro Branco, where the sand samples were collected, is located around the headquarters of the municipality of Porto Grande. The municipality of Porto Grande in Amapá is located in the center of the state 108 kilometers from the capital Macapá. The objective of this research was to make the technological analysis of material from Areal Morro Branco, Porto Grande, Amapá, by fractionation. The sand was removed from a sandy area in the municipality of Porto Grande – AP with the use of hand tools for collection. 03 (three) samples from different points with different composition were collected, due to the existence of organic matter in its composition. Sodium silicate with a concentration of 10% and bromoform was used for the breakdown of the organic matter of the sample. These procedures were determined in the dry sample in an oven for 24 hours at approximately 110ºC, and the percentage of humidity (%U), this, was also determined in the original sample. It was verified through the tests and procedures performed that the moisture of the sand of The Areal Morro Branco is influenced by the organic matter resulting from the forest areas and water resources of the surroundings of The Areal. The granulometry of the points from which the sand was collected in the sand has characteristics of angled and sub-angled grain. With the aid of bromoform density, it was feasible to realize that the sand from which the sample was collected has quartz aggregates, feldspar with a density lower than 2.89 g/cm3. It has been suggested that the sand may also have mineral derivatives of sand with density greater than 2.89g/cm3, which are Olivine and Pyroxene.
Keywords: Fractionation, sand, mineral characterization.
INTRODUCTION
Natural sand is extracted using mining methods, even for the manufacture of artificial sands. The extraction of this material occurs in a way that does not come to harm the environment. The processing of artificial sand takes place by the extraction of gravel rocks. After this extraction occurs the decrease of these rocks to obtain fine gravel. For this, processing methods such as crushing, grinding, sieving and classification are used (NASCIMENTO, 2009).
Industrial sand is generally defined as a material composed basically of silica with varied granulometry and has already undergone processing. The origin of sand formation for production is located in deposits of quartzosa, quartzite or sandstone sands (FERREIRA and DAITX, 2003).
In Brazil most of the industrial sand production is in the southern region of the country, with Santa Catarina as the state that produces the most in the southern region, but the company that produces the most in the country is in São Paulo. These companies in the South of the country supply the ceramic and glass industries respectively, causing it to arouse the interest of large companies to make investments in sand production in the region (FERREIRA and DAITX, 2003).
The processing of sand is the most important stage of all processes, as it will determine its economic use, since any sandy body can be transformed into industrial sand, from which the characteristics are linked to the original deposit (FERREIRA and DAITX, 2003).
Sand is widely used in several areas. As construction aggregates; material processing industries; sewage treatment (DNPM, 2013).
The municipality of Porto Grande in Amapá is located in the center of the state 108 kilometers from the capital Macapá. It has an estimated population in 2018 of about 21,484 people, with an area of 4,428.013 km², resulting in a population density of 3.82 inhabitants/km² with the Human Development Index – HDI of 0.64 (IBGE, 2018).
In the surroundings of the municipality are located the sand deposits, whose extraction is developed on probable river terraces. These deposits are located at a distance of 4 to 6 kilometers south of the city. The mineral that makes up these deposits is usually quartz. The sand of these deposits has fine and medium granulometry, and can be grains angled to sub-angled. The geometry, dimensions, cubation and origin of these deposits are not yet well known. They occur close to the contact of the amapaense-coastal floodplain with weathered rocks of the crystalline base. The municipality of Porto Grande has an area with more than 6,000 ha available for the extraction of sand, pebble, gravel, clay and clay (OLIVEIRA, 2010).
The Areal of the company Morro Branco, where the sand samples were collected, is located around the headquarters of the municipality of Porto Grande within a radius of 4km, at the reference point, at the geographic latitude coordinate: 78541.13 m N; longitude:463198.30 m E (MAGALHÃES et al., 2007).
With regard to the detailed characterization of the sample, it is necessary to separate it in the most appropriate way, with this is carried out the fractionation (DONAGEMMA et al., 2015).
The physical fractionation of organic matter can quantify the alterations caused by inadequate management related to soil recovery attempts. On the other hand, chemical fractionation can be applied in the evaluation of carbon content of soil hymen fractions (SANTOS et al., 2013).
In Brazil there are several studies on the physical fractionation of the soil applied for studies on soil organic matter, but these studies only quantified the organic matter that were present with silt, sand and clay. Other studies on laboratory fractionation show the dynamics of soil organic matter (MOS), and the possibility of extracting this organic matter in aggregates and those associated with sand, silt and clay (MACHADO, 2002).
In fractionation, bowl centrifue are usually used, where the granulometry obtained will be above 80% less than 2. The methods used for fractionation can be chemical or physical. In the physical method, granulometric processes can be used, such as separation using sieves, or densymmetric process, as separation with the use of dense solutions (CONCEIÇÃO et al., 2015). Before fractionation is carried out, it is necessary that there is a preparation of the sample, where it is ground and crushed to obtain a maximum size. After the comminution stage, the sample needs to undergo homogenization and quartation, in order to obtain a fraction of it. Then the material is taken to fractionation (LUZ et al., 2004).
In chemistry fractionation is characterized by the formation of several substances, resulting from the initial material (GALDINO, 2010).
In liquid-dense fractionation, at least three replicates should be used, since the light fraction has the property of being quite variable (MACHADO, 2002). Fractionation that employs dense liquid separation uses hoppers that separate the optimal volume from each fraction to be separated. The solutions that are generally used by this type of method are: Zinc chloride solution, whose density is 1.8; sodium chloride solution has a density of 2.2; sodium polytungstate, sodium metatungstate, SPT, which have a density of 2.9; lithium metatungstate, LMT, lithium heteropolytungstate, LST, density of 3.0 (LUZ et al., 2004).
It is quite common to use magnetic splitters to fractionate the sample by the wide variety of existing magnetic splitters. The most used is the Frantz isodynamic magnetic separator, a very versatile magnetic separator. The magnetic separator is composed of a vibrating trough with inclinations in its longitudinal and transverse directions, which is close to two electromagnets of adjustable current, which determine the flow density of the magnetic field. The separation of the minerals is determined by the lateral slope of the trough, the flow speed of the particles, their longitudinal inclination, the intensity of the vibration and the amount of material fed into the funnel. These longitudinal and lateral inclinations are usually found at 25 ° and 15 ° respectively, this facilitates the tableing of their mineral characteristics, but may vary in a possible refinement of the separation. The magnetic flux density of the Frantz separator can range from 0 to 20 kG, its processing is efficient in particles of particlesize between 0.833 mm to 74 μm (20 and 200 meshes) (LUZ et al., 2004).
In particles that exceed the 0.833 mm granulometry, there is a physical limit for its flow, it is necessary to increase the funnel hole, but we can have a high efficiency in smaller particles, depending on the properties of the ore (LUZ et al., 2004).
Deagglutination is a process in which the molecules to be analyzed are suspended, with the objective of disaggregating the particles where all are individualized and remain so during the analysis. For this test to occur it is necessary to use some basic substances that stabilize the pH. The substances that are generally used to perform deagglutination are: Sodium Hexametaphosphate and Sodium Pyrophosphate, but sodium silicate can also be used (DIAS, 2004).
GOAL
Make the technological analysis of material of Areal Morro Branco, Porto Grande, Amapá, by fractionation
METHOD
The sand was removed from a sandy area in the municipality of Porto Grande – AP with the use of hand tools for collection. 03 (three) samples from different points with different composition were collected, due to the existence of organic matter in its composition. Sodium silicate with a concentration of 10% and bromoform was used for the breakdown of the organic matter of the sample.
These procedures were determined in the greenhouse dry sample for 24 hours at approximately 110ºC, and the percentage of humidity (%U) was also determined, which was determined in the original sample
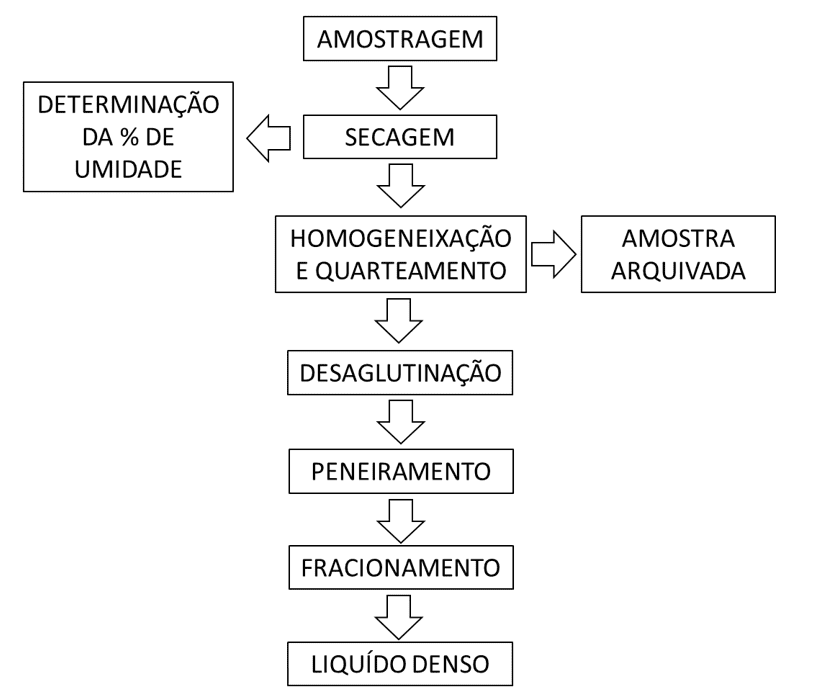
The methodology used to perform the tests is shown in Figure 1 and described below.
Figure 1 Shows the stages of the methodological procedure for performing the tests.
SAMPLING
The sand was collected at three distinct points. The first point is located N OO° 42′ 37.7”, W 051° 19′ 56.2”, with an elevation of 82 meters at sea level; the second point N 00° 42′ 31.1”, W 051° 19′ 53.6”, with elevation of 76 meters and the third point N 00° 42′ 33.4”, W 051° 19′ 58.2”, with the elevation of 72 meters in the AMorroreal Branco in the municipality of Porto Grande, Amapá, Brazil. The samples were collected at depths of 20 – 40 centimeters (Figure 2).
Figure 2 Shows the collection of material at Areal Morro Branco in the municipality of Porto Grande, Amapá, Brazil.
DRYING
The sample with 1022.49 grams (approximately 1 kilogram) was placed in the greenhouse of the mineral processing laboratory of the Federal Institute of Amapá (IFAP), at a temperature of 110°C for 24 hours. The sample, after removal, contained 936.50 grams.
HUMIDITY DETERMINATION
Moisture determination is the percentage of liquidity in a given solid. It can be determined by the equation:
U%: Humidity percentage; Mu: Moisture mass; Ms: Dry mass.
HOMOGENIZATION AND QUARTEAMENTO
The dry sample is homogenized so that it can obtain a better distribution of the material. This process is performed on a square canvas, where all the material is poured, aiming to create a tapered shape, later lifting the edges of the canvas, making the material more homogeneous. Next, the quartation is carried out, which consists of reducing the mass of the collected sample, in order to use an aliquot to perform the tests (Figure 3).
Figure 3 Shows the homogenization and quaritling assay.
DEAGGLUTINATION
12.2g of sodium silicate (Na2OSi3) dissolved in a 100ml volumetric flask of distilled water was used. Then, 500g of the sand sample was collected, it was placed in a 1L beaker and distilled water was immediately added. The sample was followed to the mechanical agitator, where 10ml of the sodium silicate solution was added to each agitation. Four agitations with duration of time were performed, respectively of 5 min, 10 min, 30 min and 1 hour. Subsequently, the sample was taken to the greenhouse.
SCREENING
The sample did not go through the comminution process, since it was the sand and was already in the appropriate size for the tests. The sand was removed from the greenhouse after the deagglutination process. For the separation of the particle size fractions, sieving was done using sieves with an opening of 8# (mesh), 16#, 35#, 60#, 80#, 100#, 200#, 250#, 325# and 400#. The retained of each sieve was used for granulometric analysis.
FRACTIONATION AND DENSE LIQUIDATION
The method chosen for the fractionation test was by dense means.
It weighed in a centrifugal bottle 1 g of each retained sieves of 60#, 80#, 100#, 200#, 250#, 325# and 400#. Using a bead, 8 ml of bromoform was added to each vial, and then the material was taken to the centrifuge where it remained for 30 minutes. The floated material was separated from bromoform by vacuum filtration. To determine the percentage of floated, the filters used were weighed before the test and after filtration and drying in the oven at 100 °C. With these values it was possible to make the difference of the masses of the filters and determined, in grams and in percentage, the amount of floated and sunk from the masses of the samples before the process.
RESULTS AND DISCUSSION
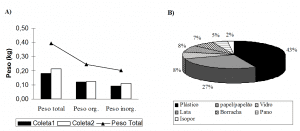
The moisture value is 8.4%, i.e. for every 100 kg of the solid-liquid mixture sample, 8.4% are water. The value of this humidity may be a consequence of the amount of water resources and large forest areas available to the surroundings of the sand and the municipality of Porto Grande (ROBACHER et al., 2013).
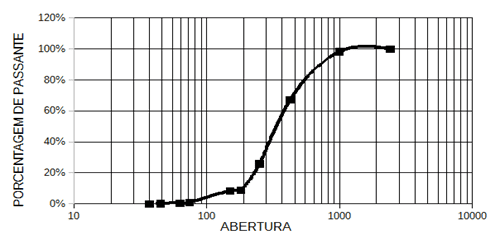
From the passer-by percentage of the sand sample. The highest percentage of passers-by is in the 8-mesh sieve, with an opening of 2400μm (Figure 4).
Figure 4. It shows the passing of the sand sample through the sieves.
This result occurs because the 8 mesh sieve, among all other sieves used, has a considerably larger opening than the grains of sand. The passage of the sample in the sieves of 200#, 250#, 325# and 400# may have been hindered by the meshes of such sieves having an opening less than the diameter of the sand grains, which on average is 0.214 mm (approximately 214. The size of the sand particles is influenced by the characteristics of the place where the material is located (VINTEM et al., 2003). This indicates that possibly the place where the sand sample was collected presents a granulometry range where most particles are greater than 214, suggesting that the sand grains of the site are angled to sub-angled (OLIVEIRA, 2010).
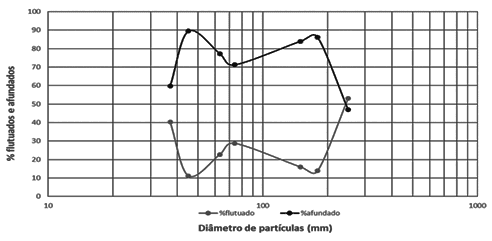
Between the percentage of floated and sunk in bromoform solution. It is verifiable that the highest percentage presented in the graph belongs to the sunken material, while the lowest percentage belongs to the floated material (Figure 5).
Figure 5. Shows the percentage of floated and sunk in bromoform solution.
Taking into account that the bromoform density is 2.89; 2.65 quartz; and that of feldspar from 2.56 to 2.76, it is possible to notice that, possibly, the floating material consists of such minerals. Knowing that the minerals of olivine and pyroxene also constitute the sand and its densities are, respectively: 3.27 to 4.37 and 3.4 to 3.5. It is infers that the sunken material is probably made up of these minerals. Therefore, these minerals have a higher density than bromoform density (DIAS, 2004).
CONCLUSION
It was verified through the tests and procedures performed that the moisture of the sand of The Areal Morro Branco is influenced by the organic matter resulting from the forest areas and water resources of the surroundings of The Areal. The granulometry of the points from which the sand was collected in the sand has characteristics of angled and sub-angled grain.
With the aid of bromoform density, it was feasible to realize that the sand from which the sample was collected has quartz aggregates, feldspar with a density lower than 2.89 g/cm3. It has been suggested that the sand may also have mineral derivatives of sand with density greater than 2.89g/cm3, which are Olivine and Pyroxene.
REFERENCES
CONCEIÇÃO, P. C. et al. Eficiência de Soluções Densas no Fracionamento Físico da Matéria Orgânica do Solo. R. Bras. Ci. Solo, v. 39, p. 490-497, 2015.
DIAS, J. A. A ANÁLISE SEDIMENTAR E O CONHECIMENTOS DOS SISTEMAS MARINHOS (Uma Introdução à Oceanografia Geológica). Portugal: Universidade de Algarve, 2004. 84p.
DNPM. Agregados para a Construção Civil. 2013. Disponível em: < http://www.dnpm.gov.br/dnpm/publicacoes/serie-estatisticas-e-economia-mineral/outras-publicacoes-1/8-1-2013-agregados-minerais >. Acesso em: 24 outubro 2018.
DONAGEMMA, G. K. et al. Avaliação do fracionamento da fração areia para a separação de solos arenosa. XXXV Congresso Brasileiro de Ciência do Solo 2015.
FERREIRA, G. C.; DAITX, E. C. A mineração de areia industrial na Região Sul do Brasil. Ouro Preto. R. Esc. Minas, v. 56, n. 1, p. 59-65, 2003.
GALDINO, L. C. Programa que quantifica o processo de cristalização fracionada e sua aplicação ao estudo de soleiras da Bacia do Paraná (Estado do Paraná). 2010. (Doutorado). Universidade de São Paulo.
IBGE. Brasil em Síntese/Amapá/Porto Grande. 2018. Disponível em: < https://cidades.ibge.gov.br/brasil/ap/porto-grande/panorama >. Acesso em: 24 outubro 2018.
LUZ, A. B.; SAMPAIO, J. A.; ALMEIDA, S. L. M. Tratamento de Minérios. Rio de Janeiro: CETEM/MCT, 2004.
MACHADO, P. L. O. A. Fracionamento físico do solo por densidade e granulometria para a quantificação de compartimentos da matéria orgânica do solo: um procedimento para a estimativa pormenorizada do seqüestro de carbono pelo solo.: Embrapa Solos 2002.
MAGALHÃES, L. A.; SOUZA FILHO, C. R.; SILVA, A. M. Caracterização geológica – geofísica da porção central do Amapá com base em processamento e interpretação de dados geofísicos. . Revista Brasileira de Geociências, v. 37, n. 3, p. 464-477, 2007.
NASCIMENTO, J. F. Estudos de areias artificiais em concreto betuminoso. Faculdade de Engenharia Kennedy,. 2009. (Especialização). Faculdade de Engenharia Kennedy Belo Horizonte.
OLIVEIRA, M. J. Diagnóstico do setor mineral do Estado do Amapá. Macapá: IEPA, 2010. 148p.
ROBACHER, L. A. et al. Plano Diretor Participativo do Município de Porto Grande, Estado do Amapá. Porta Grande: PMPG: 348p p. 2013.
SANTOS, D. C. et al. Fracionamento químico e físico da matéria orgânica de um Argissolo Vermelho sob diferentes sistemas de uso. Ciência Rural, v. 43, n. 5, p. 838-844, 2013.
VINTEM, G.; TOMAZELLI, L. J.; KLEIN, A. D. F. O Efeito do Tamanho de Grão de Areia No Processo de Transporte Eólico dos Campos de Dunas Transgressivas do Littoral do Estado de Santa Catarina, Brasil. II Congresso sobre Planejamento e Gestão das Zonas Costeiras dos Países de Expressão Portuguesa. IX Congresso da Associação Brasileira de Estudos do Quaternário. II Congresso do Quaternário dos Países de Língua Ibéricas. Recife PE 2003.
[1] Mining Technique. Institute of Basic, Technical and Technological Education of Amapá (IFAP).
[2] Mining Technician. Institute of Basic, Technical and Technological Education of Amapá (IFAP).
[3] Biologist, PhD in Theory and Behavior Research, Professor and researcher of the Degree Course in Chemistry of the Institute of Basic, Technical and Technological Education of Amapá (IFAP).
[4] Biomedical, PhD in Topical Diseases, Professor and researcher of the Medical Course of Macapá Campus, Federal University of Amapá (UNIFAP).
[5] Theologian, PhD in Psychoanalysis, researcher at the Center for Research and Advanced Studies – CEPA.
[6] Materials Technology. Master in Mechanical Engineering, Professor and researcher of the Degree Course in Chemistry of the Institute of Basic, Technical and Technological Education of Amapá (IFAP).
Sent: March, 2020.
Approved: March, 2020.